Neisseria gonorrhoeae LOS Decreased NF-ĸB and IRF3 Expression but Had No Effect in IFN-β Expression and HIV Replication on Naïve-HIV Lymphocytes
Aннотация
Background:Neisseria gonorrhoeae (N. gonorrhoeae) is a gram-negative bacterium with a lipooligosaccharides (LOS)-based cell membrane. LOS is considered to supress HIV-1 replication by downregulating NF-κB, IRF3, and IFN-β. The aim of the study:To explore the effect of N. gonorrhoeae LOS on HIV replication in lymphocytes of naive HIV patients, as indicated by NF-κB, IRF3, and IFN-β expressions. Materials and methods: Naïve-HIV lymphocyte culture was divided into 5 groups. The treatment groups were exposed to N. gonorrhoeae LOS with doses ranging from 50 ng/ml, 100 ng/ml, and 200 ng/ml, respectively. The positive control group was exposed to 300 µM of tenofovir as the standard antiviral treatment, while the negative control group received only standard medium. At 24 hours post-treatment, the level of HIV p24 was determined using ELISA and the expressions of NF-κB, IRF3, and IFN-β was measured by flowcytometry. Results: The mean expression of NF-κB and IRF3 were significantly different among groups (p=0.025; p=0.020), while the expression of IFN-β and the level of p24 did not differ significantly (p=0.051; p=0.068). There was a strong and significant positive correlation between LOS concentration and NF-κB and IRF3 expression (r=0.704, p=0.003; r=0.759, p=0.001;). Conclusion: Exposure of lymphocytes from a naïve HIV patient to N. gonorrhoeae LOS significantly affected the expression of NF-κB and IRF-3.
Ключевые слова: Lipooligosaccharides, Nuclear factor-kappa beta, Interferon Regulatory Factor 3, Interferon-β, Human immunodeficiency virus
К сожалению, текст статьи доступен только на Английском
Introduction.Human immunodeficiency virus (HIV) is a retrovirus that infects, destroys, or impairs the function of cells of the immune system. As the infection progresses, the immune system weakens, making patients more susceptible to other infections [1]. As HIV progresses, the acquired immunodeficiency syndrome (AIDS) may develop within 5 to 10 years. The prevalence of HIV/AIDs has increased in the last decade and requires better methods to improve the quality of life of people living with HIV-AIDS by reducing the progression of the disease [2]. The United Nations Programme on HIV/AIDS (UNAIDS) reported an estimated global prevalence of 39 million, whereas it is estimated that 570,000 adults and children in Indonesia are living with HIV and AIDS [3, 4].
The WHO reported that HIV co-infection with Neisseria gonorrhoeae (N. gonorrhoeae) markedly deteriorated the condition of HIV patients [5]. The combination of HIV and other STIs has a particularly negative impact on the health of adolescent and young adult populations [6]. Those with sexually transmitted illnesses (STDs) are generally at a higher risk of developing HIV since an STD can cause a sore or a breach in the skin, making it easier for HIV to enter the body [7]. Interestingly, a cohort study in Kenya reported that female sex workers who contracted N. gonorrhoeae shortly before or during HIV infection had stronger and better HIV-specific immunity than those who were not co-infected or infected with other sexually transmitted infections (STIs).This clinical observation warrants a possible relationship between N. gonorrhoeae infection and HIV resistance [8].
Neisseria gonorrhoeae is a gram-negative bacterium with a lipooligosaccharide (LOS)-comprising cell membrane, which ought to decrease HIV-1 replication in the natural immunity [9]. N. gonorrhoeae outer membrane LOS is composed of three oligosaccharide chains connected to a lipid A core, and because of the lack of a repeating polysaccharide O-side chain, is structurally unique from lipopolysaccharide of other Gram-negative bacteria [10]. LOS induces natural immunity through Toll Like Receptor-4 (TLR4) on dendritic cells (DC) and macrophages as well as non-immune cells, such as fibroblasts and epithelial cells. Stimulation of TLR4 by LOS induces the release of proinflammatory cytokines to activate the natural immune response, particularly lymphocytes. TLR4 activates Nuclear factor-kappa beta (NF-κB) after recognizing pathogen infection or tissue damage [11], which is required for expression and subsequent signal transduction, and further affects various cellular processes, including the inhibition of HIV replication [12]. This function is performed through the MyD88-independent pathway associated with the induction of dendritic cell maturation, and activation and translocation of NF-κβ transcription factor into the nucleus [13].
Induction of TLR4 by LOS also activates the recruitment of various TIR domain-containing adaptor molecules and induce particular immune responses to eliminate invading pathogens. Several adaptors have been identified in mammals, including TIR domain-containing adaptor-inducing interferon-β (TRIF) and TRIF-related adaptor molecule (TRAM). TRIF is unique to TLR4-mediated signalling pathways. It activates Interferon Regulatory Factor 3 (IRF3) and NF-κB and induces the production of type I IFN and inflammatory cytokines, leading to direct killing of invading pathogens [14]. IRF3 plays a major role in upregulating endogenous SAMHD1 expression, a type I interferon inducible host innate immunity restriction factor that inhibits the early steps of the viral life cycle. SAMHD1 blocks the replication of HIV-1 in myeloid lineage cells and functions in hydrolyzing deoxynucleotide triphosphate (dNTP) which further inhibits the reverse transcriptase (RT) of HIV-1 [15].
Our preliminary in silico study showed that N. gonorrhoeae LOS showed the strongest binding affinity for TLR4 compared to E. coli LPS and laminaran, an active compound of the brown algae LPS. When viewed from the attachment side, these three ligands were found to bind to the same region as eritoran, the control ligand. These results indicated that N. gonorrhoeae LOS was capable of inducing inflammation via TLR4, although its binding was not as good as that of eritoran. In this study, we aimed to explore the effect of N. gonorrhoeae (ATCC 49226) LOS administration toward HIV replication, as indicated by IFN-β, IRF3, and NF-ĸB expression, and HIV p24 level. These might shed light into the interplay of HIV-STI and its implication of future treatment strategies.
Methods
Study Design
This study was an experimental laboratory study with a post-test only control group design method. This study was conducted to observe the effect of N. gonorrhoeae (ATCC 49226) LOS in suppressing HIV replication through the NF-κβ, IRF3, and IFN-β pathways in lymphocytes of naïve HIV patients. In this study, the expression of IFN-β, NF-ĸB, and IRF3 was measured using the flowcytometry. The level of p24 HIV-1 virus was determined using ELISA. This research protocol has been approved by the Ethics Committee of dr. Saiful Anwar Hospital, Malang, East Java, Indonesia, with ethics number 400/024/K.3/302/2020 dated January 24th, 2020.
Study Participants and Samples
Recruitment of study participants was carried out by screening patients and providing explanations. All participants filled out informed consent forms. Demographic data was collected by interviews to explore risk factors for HIV infection. History taking, physical examination, and laboratory tests were performed to determine disease stage, opportunistic infections, and other comorbidities.
Blood samples from consenting naïve HIV patients, with confirmed diagnoses according to the WHO criteria, were collected. Twenty millilitres of venous blood were collected in an EDTA vacutainer tube. The tubes were labelled and processed immediately for plasma and monocyte isolation.
Lymphocyte Isolation and Culture
A 15 ml centrifugal bottle (Terumo) was prepared and filled with Ficoll-Hipaque d=1.077 g/ml. The diluted blood sample was carefully layered onto the Ficoll-Hipaque media solution in a volume ratio of 1:1, then centrifuged (Biosan LMC-3000) at room temperature at 1000 rpm for 30 minutes. A sterile pipette was used to remove the upper layer of plasma and platelets, leaving the mononuclear cell (PBMC) layer intact at the interface. The PBMC ring was carefully removed with a micropipette, placed in a new 15 mL centrifuge bottle, and rinsed with 10 ml PBS, before being centrifuged at room temperature for 10 minutes at 1200 rpm, which was repeated twice. PBMC pellets were resuspended with growth medium in a Petri dish and incubated at 37.5% CO2 for 2 hours. Non-attached cells (lymphocytes) were collected and resuspended in growth medium.
Flowcytometry Examination for the Detection of Surface Markers and Apoptotic Markers of Lymphocytes
Each lymphocyte sample received 20 µl of BD CD45+/CD3+/CD4+/CD8+ antibody cocktail. To detect apoptosis, a solution of 5 µl of FITC annexin V and 10 µl of propidium iodide (PI) were combined with 400 µl of binding buffer. Each of these suspensions was then mixed with the antibody-treated cell suspension and incubated for 20 minutes. The cells were rinsed with 500 µl staining buffer and centrifuged at 2500 rpm (350 g). The cell pellet was mixed with 500 µl of fixation buffer, incubated at 4ᵒC for 20 minutes, and rinsed with 500 µl staining buffer, followed by centrifugation at 2500 rpm (350 g). The pellet was mixed with 500 µl of fixation buffer and incubated at 4ᵒC for 20 minutes. After centrifugation at 2500 rpm, the pellet was rinsed with 1 ml of staining buffer and re-centrifuged. Finally, 300 µl of staining buffer was added to the pellet and the suspension was homogenized by vortexing before being transferred to a 5 ml tube for analysis using BD FACS Melody Flowcytometry.
Neisseria gonorrhoeae (ATCC 49226) Bacterial Culture
Neisseria gonorrhoeae (ATCC 49226) was obtained as a colony culture from BBLK Surabaya. Gram staining and the oxidase test were used to identify the bacteria, which were subsequently cultivated on modified Thayer Martin agar and incubated at 37°C for 48 hours with 3-10% CO2. Subcultures were performed until a minimum volume of 500 ml was attained, which was sufficient for LOS extraction and purification.
LOS Extraction and Purification of Neisseria gonorrhoeae
Lipooligosaccharide (LOS) was extracted from N. gonorrhoeae (ATCC 49226) using a modified hot-phenol water method. The process involved centrifugating 500 ml of bacterial culture at 15,000 g for 1 hour to obtain pellets, which were then suspended in 10 ml of deionized water at 72-75°C. Hot liquid phenol (88%) was added, and the mixture was vortexed, incubated, and centrifuged. After removing the protein interface, the phenol layer was discarded, and the aqueous layer was dialyzed to remove residual phenol in 4°C water for 48 hours, followed by overnight dialysis with Na-EDTA. Finally, LOS was precipitated using 95% ethanol and sodium acetate, left at -20°C overnight, and centrifuged. The resulting pellets were resuspended in 1 ml of water and stored at -20°C.
Identification of Neisseria gonorrhoeae LOS
Western blotting was used to identify LOS. For the SDS-PAGE, a 48% polyacrylamide gel was prepared, and samples with equal protein amounts were loaded into the wells. Electrophoresis was run at 100 V for 1-2 hours. The gel was moistened with transfer buffer and arranged in a transfer cassette for protein transfer onto the nitrocellulose membrane. The membrane was washed, molecular weight bands were marked, and then incubated in blocking solution for 1 hour at room temperature. The membrane was then incubated with primary antibody solution in 1x TBST and 1% BSA for 1 hour at room temperature, followed by three 10-minute washes with 1x TBST. Next, the secondary antibody solution in 1x TBST and 1% BSA was added for 1 hour at room temperature, followed by additional washes. For protein detection, the membrane was incubated with ECL substrate for 1-5 minutes, placed between plastic layers, and exposed to autoradiographic film. The film was then aligned over the blot to visualize the molecular weight ladder (Fig. 1).
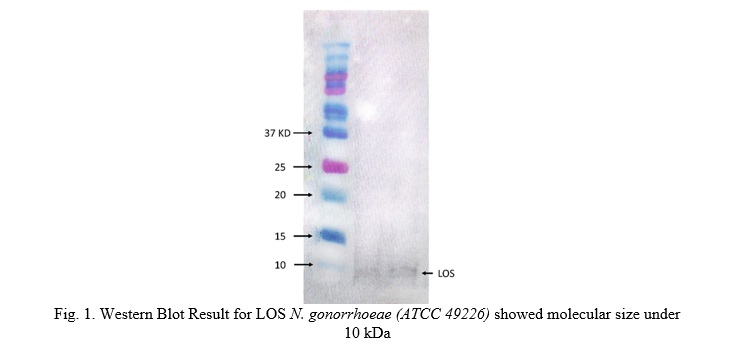
Exposure of Neisseria gonorrhoeae LOS to Lymphocytes culture
The suspension of N. gonorrhoeae LOS was produced in a 0.5% DMSO solvent at a concentration of 1000 µM. Working concentrations of 50, 100, and 200 ng/ml was prepared using standard culture media (RPMI 1640, 10% FBS, 1% penicillin/streptomycin) as diluent. The suspension of medium and treatment agent was then introduced to the lymphocyte culture.
The lymphocytes were divided into 3 treatment and 2 control groups. The treatment groups, T1, T2, and T3, were exposed to LOS N. gonorrhoeae at concentrations of 50 ng/ml, 100 ng/ml, and 200 ng/ml respectively. The positive control (CP) group was exposed to the standard HIV treatment of 300 µM tenofovir disoproxil fumarate (tdf), while the negative control group did not receive any exposure.
The lymphocytes in all groups were incubated in culture medium containing RPMI 1640, 10% FBS, 1% penicillin, and streptomycin at 37°C for 24 hours, before determining levels of p24, NF-κB, IRF3, and IFN-β expression.
Flowcytometry Examination for Determination of NF-κB, IRF3, and IFN-β Expression
Cells from each well were harvested separately and centrifuged at 220-350 g at 4°C for 5 minutes. The pellets were washed with sterile PBS and centrifuged at 13,000 rpm for 10-20 seconds. Next, they were resuspended in PBS to achieve 10 × 10^6 cells/ml, incubated with a blocking reagent for 10 minutes, and resuspended in 50 ml PBS to remove antibodies. After centrifugation at 1,000 rpm for 5 minutes, the pellets were resuspended in FCM fixation buffer, incubated for 30 minutes at room temperature, washed with PBS, and centrifuged again. Cold FCM permeabilization buffer was added, vortexed, incubated for 5 minutes, and centrifuged. Then, the pellets were resuspended in FCM wash buffer to 10 × 10^6 cells/ml, followed by the addition of 1 µg antibodies per tube (100 µl of cell suspension). After incubating for 15-30 minutes, excess antibodies were washed off with PBS and centrifuged. Cells were finally resuspended in 500 µl of 1% paraformaldehyde. The suspension was stored in a dark room for 24 hours, then transferred to a 5 ml tube for analysis by BD FACS Melody Flow Cytometry.
ELISA Examination to Measure HIV p24 Levels
Two-hundred microliters of each control and specimens were added to the respective wells and incubated at 37ᵒ C for 60 minutes. After washing, 100 ml of streptavidin HRP conjugate was added to each well and incubated for 30 minutes at room temperature. The conjugate was removed and washed, followed by the addition of 100 ml of substrate and incubated at room temperature for 30 minutes. The reaction was stopped by adding 100 ml of stop. The optical density was immediately read at 450 nm using the microplate reader.
Data Analysis
The data were analyzed using SPSS version 24 for Windows, with a significance level set at 0.05 and a 95% confidence level. The comparative hypothesis testing steps included the Kolmogorov-Smirnov normality test and Levene's test for data homogeneity. As the NF-κB and IRF3 levels were not homogeneously distributed, they were analysed using the Kruskal-Wallis test, followed by a post hoc analysis. The p24 and IFN-β were analysed using One-way ANOVA for the mean difference test.
Results
Lymphocyte Surface Markers
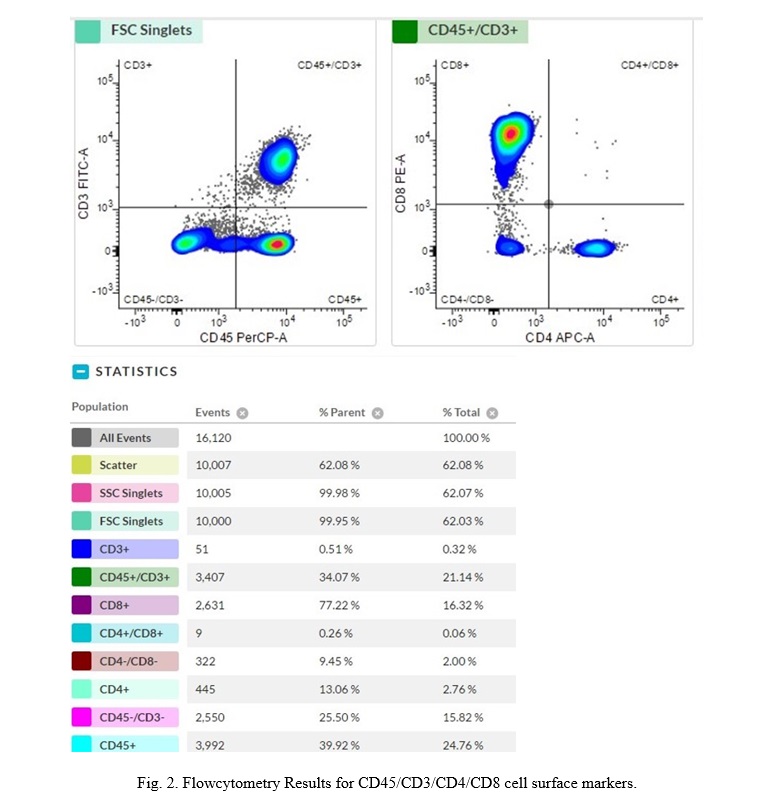
After isolation of PBMC, the lymphocytes were cultured. The identification of cell surface markers was carried out using flowcytometry to ensure the availability of lymphocyte (CD45+/CD3+/CD4+/CD8+), as shown in Fig. 2.
Effect of Neisseria gonorrhoeae LOS Exposure on Lymphocyte NF-κB Expression
The expression of NF-κB in each treatment group was examined using the flowcytometry after exposure to N. gonorrhoeae LOS. The NF-κB expression in the treatment group is shown in Fig. 3.
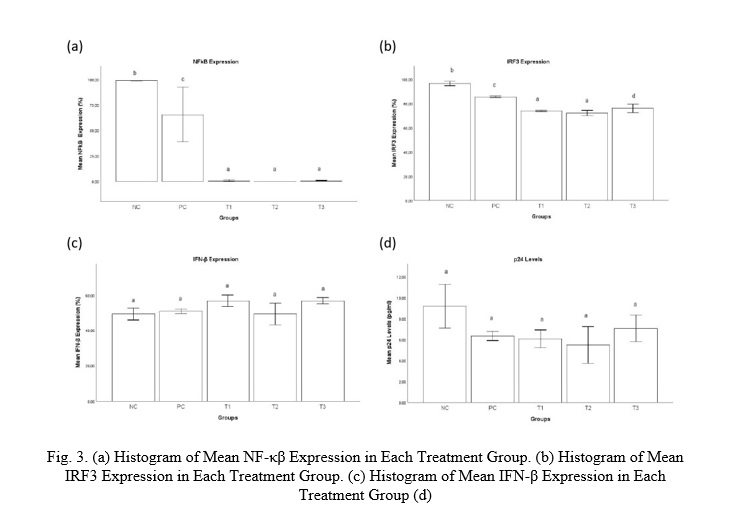
The treatment groups (N. gonorrhoeae LOS) had considerably decreased NF-κB expression compared to the negative and positive control groups. The mean expression of NF-κB among the groups had significant differences (p=0.025). The Pearson correlation test showed a strong and significant positive correlation between LOS dose and NF-kB expression (r=0.704; p=0.003)
Effect of Neisseria gonorrhoeae LOS Exposure on Lymphocyte IRF3 Expression
Interferon Regulatory Factor 3 (IRF3) expression in each treatment group was examined using the flowcytometry after exposure to N. gonorrhoeae LOS. The IRF3 expression in the treatment group is shown in Fig. 3.
The IRF3 expression in all the treatment groups (N. gonorrhoeae LOS) was considerably lower compared to negative and positive control groups, and there was a significant difference in mean IRF3 expression between LOS dosages 100 and 200 ng/ml (p=0.020). The Pearson correlation test showed a strong and significant positive correlation between LOS dose and IRF3 expression (r=0.759; p=0.001).
Effect of Neisseria gonorrhoeae LOS Exposure on Lymphocyte IFN-β Expression
IFN-β expression in each treatment group was examined using the flow cytometry method after exposure to N. gonorrhoeae LOS. The mean of IFN-β expression in the treatment group is shown in Fig. 3.
Despite our premise that LOS would enhance IFN-β expression, all treatment groups had lower IFN-β expression than the negative control group, however the difference was not significant (p = 0.051).
Effect of Neisseria gonorrhoeae LOS Exposure on Lymphocyte p24 Level
The p24 level in each treatment group was examined using the ELISA method after exposure to N. gonorrhoeae LOS. The mean of p24 level in the treatment group is shown in Fig. 3.
It was found that the p24 level in all treatment groups was lower compared to negative control group, however the differences among treatment groups were not significant (p=0.068). The highest p24 level was found in the negative control group, while the lowest was found in treatment group 2 with LOS dose of 100 ng/ml.
Discussion
Flowcytometry Results Lymphocyte Surface Markers
The results of lymphocyte culture flow cytometry showed that 99.95% of cells were lymphocytes consisting of 0.51% CD3+, 77.22% CD8+, 13.06% CD4+ and 39.92% CD45+ cells. Previous studies stated that T lymphocytes in healthy people contain approximately 65% CD4+ and 35% CD8 T lymphocytes. During HIV infection, CD4+ T cells decline and malfunction, whereas CD8 T lymphocytes grow, resulting in a CD4+/CD8 imbalance. People living with HIV with a low CD4/CD8 ratio show more inflammation and immunosenescence. Several researches have looked into whether the CD4/CD8 ratio predicts the risk of mortality in people living with HIV. Some studies have identified a substantial relationship between a low CD4/CD8 ratio and an increased risk of mortality [16].
Neisseria gonorrhoeae LOS strongly reduced lymphocytes NF-ĸB expression of naive HIV patients
A previous study stated that LOS stimulation on monocyte-derived macrophage (MDM) will activate several transcription factors including NF-ĸB and AP-1. These active transcription factors will induce several host factors such as proinflammatory cytokines, chemokines and host cell protective factors [17]. In this study LOS of N. gonorrhoeae (ATCC 49226) strongly reduce lymphocytes NF-ĸB expression, potentially through several mechanisms such as inhibiting kinase activity, preventing nuclear translocation of p65, inhibiting nuclear p65 activation, and post-translational modification. [18]. The reduction of NF-ĸB expression was also identified in the positive control group that treated with standard therapy.
It is known that NF-ĸB acts as a major inflammatory mediator in immune cells. Regulation of NF-ĸB activation is vital in various inflammatory diseases, therefore approaches related to inhibition of NF-ĸB signaling pathway have been widely investigated as therapeutic targets by inhibition of key components of the canonical NF-κB pathway to block its downstream targets or upstream stimulators Several methods to inhibit NF-ĸB have been proposed, such as increasing the selective amount of IKK inhibitors to prevent Ikβα phosphorylation. The therapeutic approaches targeting NF-κB signaling through various points involving the receptor, IKK complex, gene transcription, post-translation or at any steps in between. This has been accomplished using small molecules, peptides, oligonucleotides, monoclonal antibodies (mAbs), proteasome inhibitors, nuclear translocation inhibitors, DNA binding inhibitors, and small interfering RNA (siRNA) [19, 20].
Neisseria gonorrhoeae LOS reduced lymphocytes IRF3 expression of naïve HIV patients
The activation of TLR4 is expected to phosphorylate IRF3 which will in turn induce the production of type I IFN. Therefore, in this study the exposure to N. gonorrhoeae LOS in the treatment group is expected to increase IRF3 expression in lymphocytes, however the result of this study showed the reduction of IRF3 expression. The possible factor that causes low IRF3 expression is the inactivation of the IRF3 transcription factor due to autoinhibition after exposure to N. gonorrhoeae LOS. Excessive IRF3 activation causes hyperphosphorylation of some serine in the signal response domain (SRD) which causes autoinhibition [21].
Viral inflammation contributes to pathogenesis and mortality during virus infections. IRF3, a critical component of innate antiviral immune responses, interacts with pro-inflammatory transcription factor NF-κB, and inhibits its activity [22]. Following viral infection, the virion constituents, particularly viral nucleic acids, are sensed by pathogen recognition receptors (PRRs). These receptors use adaptor proteins to stimulate a signal transduction pathways that sequentially triggers the next molecule downstream in the pathway ultimately culminating in the activation of IRF3 [23].
As the key component of antiviral immunity, IRF3 activity must be carefully regulated to ensure a strong immune response to viral infection. However, IRF3 is implicated in the generation of inflammatory cytokines, which can be harmful to the host. As a result, the ability to quickly activate and deactivate IRF3 is critical to maintaining immunological homeostasis. The body ensures that IRF3 activity returns to basal levels by degrading the IRF3 protein, thereby avoiding severe inflammation. A mechanism has been documented whereby apoptotic caspases break down IRF3, resulting in its destruction by the proteasome [24].
This study predicted the role of the MyD88 pathway in the low expression of IRF3 in the treatment group. TLR4 stimulation will activate two downstream pathways, namely the MyD88 dependent pathway mediated by the TIRAP adapter protein which ultimately activates NF-κβ, as well as the independent MyD88 pathway mediated by the TRAM adapter protein which stimulates the factor phosphorylation. Transcription of IRF3 stimulates the production of type I IFN sequentially [21]. In this study, the pathway that is expected to be activated is the independent MyD88 pathway which increases the IRF3 phosphorylation.
Neisseria gonorrhoeae LOS did not increase IFN-β expression in lymphocytes of naïve HIV patients
In contrast to IRF3, the IFN-β expression in the treatment group (N. gonorrhoeae LOS) did not differ from that in the negative control group. It was possible due to the use of purified LOS since whole-cell bacterial challenge is more stimulatory than challenge with purified LOS. The previous study demonstrated that the carbohydrate structures of LOS play an important role in disease pathogenesis. Although alterations in the carbohydrate moiety of LOS do not impact the production of most cytokines by human monocytes, modifications to the oligosaccharide could alter gonococcal–host cell interactions. This is because the lipid A portion of the LOS is responsible for immunological activation [25].
There is increasing evidence that the overall outcome of IFN-β responses can be advantageous or deleterious to the host, depending on the timing, quantity, and source of IFN-β production, as well as the unique biological environment. Further research is needed to understand the spatiotemporal induction of the various type I IFN subtypes in vivo, as well as the cell types responsible for type I IFN production. A significant study has created RNA-lipoplexes containing neoantigens or endogenous self-antigens, which resulted in rapid and robust IFN production [26].
Neisseria gonorrhoeae LOS did not reduce the level of p24 in supernatant of lymphocytes culture of HIV-naïve patients
This result differed from the earlier work which used N. gonorrhoeae MS11-derived strains that successfully suppresses HIV-1 replication [27]. It is important to consider that the gonococci are unique as their biological niche overlaps with the primary site of HIV exposure in both the male and female urogenital tracts. Taken together, the studies to date suggest that N. gonorrhoeae stimulates HIV-1 replication by upregulating host cellular activation pathways that lead through NF-ĸB in the infected CD4+ T cell, while simultaneously stimulating anti-viral immunity by eliciting a potent IFN-α; the cumulative result of these opposing effects will ultimately determine the outcome in a co-infected individual [28].
Conclusion. It could be concluded from this study that exposure of LOS from N. gonorrhoeae (ATCC 49226) to lymphocytes of naïve HIV patients had significant effects on reducing NF-ĸB and IRF3 expression, but had no effect on IFN-β and p24 expression levels. These findings may help to understand the immunopathogenesis of HIV co-infections and the use of LOS from a non-pathogen source as a replacement to suppress inflammatory signalling pathways, thereby preventing viral inflammation.
Financial support
This project was funded by the grant of the Research and Community Service Unit, Faculty of Medicine Universitas Brawijaya, with grant number 3125/UN10.F08/PN/2021.





















Список литературы
Список использованной литературы появится позже.