Impact of Aerobic Exercise on Expression of OPN and OPG Genes and Bmal1 Protein in the Bone Tissue of Diabetic Rats During Two Phases of Light and Dark Circadian Rhythm
Aннотация
Background: Diabetes is a major global disease with a high mortality rate.The impact of exercise on bone tissue protection in diabetic rats during circadian cycle phases is unknown. This study examined the effects of endurance training on genes in diabetic rats during light and dark circadian rhythm phases. The aim of the study:The present study investigated the impact of endurance training on insulin resistance and gene expression in diabetic rats during circadian rhythm phases. Materials and methods: In this study, 18 NMRI mice were randomly assigned to 6 groups: healthy control in the dark, healthy control in the light, diabetic control in the dark, diabetic control in the light, and diabetic exercise in the dark. Then, they developed diabetes after being being fed a high-fat diet and receiving a single injection of streptozotocin. The 8-week endurance training program consisted of 5 days per week of Vmax 50–60%. Blood and bone tissue were removed following anesthesia. Bone tissue was examined for the expression of OPN and OPG genes, Bmal1 protein expression, and insulin resistance index. Results: In diabetic rats, eight weeks of endurance training significantly reduced insulin resistance (P=0.005) and significantly increased the expression of the Bmal1 protein (P=0.009). OPG and OPN variables did not significantly differ between the diabetic and control exercise groups during the changes in the two phases of darkness and light (P>0.05). Conclusion: Endurance training may improve insulin sensitivity in diabetics by enhancing antioxidant system performance and controlling circadian activity. Further research is needed to determine the effects on genes in bone
К сожалению, текст статьи доступен только на Английском
Abbreviations List | |
NMRI Mice | Naval Medical Research Institute Mice |
ANOVA | Analysis Of Variance |
OPN | Osteopontin |
OPG | Osteoprotegerin |
Bmal1 Protein | Brain And Muscle Arnt-Like Protein-1 |
T2DM | Type 2 Diabetes Mellitus |
STZ | Streptozotocin |
HFD | High-Fat Diet |
QPCR | Real-Time PCR and Quantitative PCR |
CD-ZT3 | Light Phase Diabetic Control Group |
CD-ZT15 | Dark Phase Diabetic Control Group |
TD-ZT3 | Light Phase Diabetic Exercise Group |
CH-ZT3 | Light Phase Healthy Control Group |
CH-ZT15 | Dark Phase Healthy Control Group |
CD-ZT3 | Light Phase Diabetic Control Group |
PKB | Protein Kinase B |
MAPK | Mitogen-Activated Protein Kinase |
ERK | Kinases Regulated by Extracellular Messages |
T2D | Type 2 Diabetes |
ROS | Reactive Oxygen Species |
RNS | Reactive Nitrogen Species |
NADPH | Nicotinamide-Adenine Dinucleotide Phosphate |
PI3KAKT | Phosphatidylinositol 3‑Kinase/Protein Kinase B |
HIIT | High-Intensity Interval Training |
Introduction. Diabetes is a global chronic disease causing significant negative impacts on lives, families, and communities. It is one of the top ten causes of death for adults and is expected to affect 700 million people globally by 2045 [1-5]. Diabetes can develop a number of complex systemic complications due to improper management and care, including diabetic retinopathy, diabetic nephropathy, and diabetic neuropathy [6, 7, 8]. Osteoporosis is also brought on by diabetes [9], which is a condition that results in a decline in bone density and the breakdown of the structure of skeletal tissue, both of which increase the risk of fracture [10]. Bone formation and resorption are essential for maintaining active metabolic tissue. When imbalanced, bone density decreases, leading to more bone loss through reabsorption than creation. Type 2 diabetes affects skeletal tissue and bone metabolism, with poor blood sugar control impacting both formation and resorption. Important bone formation indicators include osteopontin (OPN) and osteoprotegerin (OPG) [11]. OPN is a protein that is expressed in a variety of cells, including lymphocytes, smooth muscle cells, endothelial cells, macrophages, and osteoblasts. As a result, it contributes to the pathology of several inflammatory diseases, including cardiovascular condition and allergic disease [12]. Additionally, obese individuals have higher plasma levels of OPN or fat tissue OPN gene expression [13]. The main risk factors for the emergence of insulin resistance, type 2 diabetes, and diabetes-related complications like small vessels and liver diseases are the same elevated OPN levels in obesity [14]. OPG, a secreted glycoprotein from osteoblast generation cells, binds to the nuclear factor kappa-B receptor activator, neutralizing its effect and preventing bone resorption. It inhibits osteoclast precursor differentiation into mature osteoclasts [15].
the circadian clock system in mammals is crucial for maintaining homeostasis [16]. Investigations have shown that circadian disruption is associated with metabolic disorders and type 2 diabetes [17], Exercise can help with metabolic issues linked to circadian rhythm [17]. The body's physiological and biochemical processes must be regulated by circadian rhythm. Animals' circadian rhythms are tightly regulated by the suprachiasmatic nucleus of the brain's hypothalamus [18]. The optical signals that pass through the optic nerves set the clock and different behaviors. Alternately, the circadian oscillator transcription factors Bmal1 (brain-muscle Arnt-like 1) and CLOCK (circadian locomotor output control kaput), which control cellular functions, are expressed by the central nervous system as well as nearly all peripheral tissues and cells. They modify [18]. Cell-autonomous circadian rhythms are controlled at the molecular level by coupled transcriptional and translational feedback loops. Bmal1 and CLOCK proteins make up this feedback's favorable arm. The processes required to preserve glucose homeostasis are regulated by this internal clock system [17]. Bmal1, one of the key clock genes, is expressed under the control of messenger, metabolic, and epigenetic regulators [19]. There is evidence that T2DM can be improved by changing eating or exercise schedules [20]. Tahara et al. reported that the right time is effective in preventing obesity, diabetes, and cardiovascular diseases and that the physiological reactions to stress and sports activity are dependent on the time of physical exercise [21]. Dalbram et al. have demonstrated that physical activity increases the amount of Bmal1 protein in muscle tissue and that activity during the dark phase reduces obesity without altering insulin sensitivity or glucose homeostasis [17].
The expression of OPN and OPG genes increases in obesity [13]. These elevated levels in obesity are risk factors for the development of insulin resistance and type 2 diabetes [14], so we will be looking at them in this study.
Due to the lack of research on the effect of endurance exercise on bone indices as a mediator of the effect of diabetes on osteoporosis and also the highest level of expression of the OPN and OPG genes in bone tissue compared to other tissues [12, 22], the bone tissue of diabetic rats was selected as the target tissue in this research.
The aim of the study. To compare the effects of an endurance training program during light and dark phases on bone indices in diabetic rats, considering the significant impact of mechanical pressures on bone regeneration and the influence of circadian rhythm on bone health.
Materials and Methods. This experimental study utilized simple random sampling and laboratory procedures. Eighteen adult male NMRI rats were induced with diabetes using a high-fat diet and low-dose Streptozotocin (STZ) injection [23]. The study aimed to investigate the effects of diabetes on bone indices. STZ was made in a citrate buffer with a pH of 5 every day, then injected within 30 minutes. Rats were randomly divided into four diabetic groups based on weight homogenization [24] and healthy control groups at the same time five days after STZ injection if their fasting glucose level was greater than 126 mg/dl. They received a regular diet and STZ solution and were purchased along with the diabetic group. The Razi Rad Company produced transparent polycarbonate cages that were 30 cm long, 30 cm wide, and 30 cm high and were used to house mice in three sets. The environment had a temperature of 22±2°C, a humidity level between 45% and 55%, and a light-to-dark cycle of 12:12 hours. Standard pellet food and water were freely available to the samples throughout the research.
At the end of the adaptation week, the fourth and eighth weeks of the main training, the maximum speed test was taken from all groups. Using Vmax measurements taken at the start and fourth weeks of training, training intensity in the groups was adjusted by training within the desired range. Rats from various groups were positioned on a treadmill and trained for 5 minutes at a speed of 6 m/min. The speed increased to 2 meters per minute every two minutes when the mice were unable or unwilling to work any longer. The rats' maximum speed was recorded as their final running speed, and to avoid any disruptions and their impact on the rats' performance and the research's findings, the maximum speed was measured for each group once every hour that corresponded to that group [25, 26].
The training times were three hours after the onset of light at 9 am (ZT3) and three hours after the onset of darkness at 6 pm (ZT12) to determine the training time of the day.he exercise groups worked out at two different times during the day one week after ensuring the induction of diabetes [11]. The rats were gradually introduced to the treadmill for a week before the primary training program to help them get used to it after the onset of darkness or at 9 PM (ZT15) [11]. The development and implementation of the training program took place in training groups, with each session lasting 80 minutes and being conducted at a moderate intensity (Vmax 50-60%). Weekly assessments of weight and blood sugar levels were made throughout the 8-week exercise program, which included five sessions per week [25].
The rats were moved to the sampling room 48 hours after the eight-week training period ended, and ketamine (75 mg/kg of body weight) and xylazine (10 mg/kg of body weight) were intraperitoneally administered. Their serum samples were separated after blood samples were drawn directly from the heart and centrifuged for five minutes at a speed of 5000 rpm. At the same time, pancreatic tissue was taken and stored in a -70 freezer for further analysis. Each group was examined during its specific training hour because multiple time points are required to detect changes in the phase or amplitude of the molecular clock [16].
In order to determine the insulin resistance index (HOMA-IR) of blood glucose using the glucose oxidase and spectrophotometry method (Pars Azmoun, Iran) and insulin using the species-specific ELISA method (Bioassay Technology, China) with extra-test accuracy of 3.4% and intra-test accuracy of 4.3%, insulin resistance was calculated using Equation 1:

When comparing diabetic subjects to healthy rats, the HOMA-IR value either increased or decreased, indicating an increase or decrease in insulin resistance, respectively.
In order to evaluate changes in the expression level of OPG and OPN genes, real-time PCR was performed using the qPCRTM Green Master Kit for SYBR Green I (Jena Bioscience, Germany) on a Lightcycler Detection System (Roche, USA). Relative expression levels of the OPG and OPN transcripts were compared to those of rat GAPDH as a housekeeping gene. Specific sets of primers (Bioneer, South Korea) designed for this study were: OPG (GenBank: NM_008764): 5′- CGGAAACAGAGAAGCCACGCAA -3′ and 5′- CTGTCCACCAAAACACTCAGCC -3′ and OPN (GenBank: NM_009263): 5′- GCTTGGCTTATGGACTGAGGTC -3′ and 5′- CCTTAGACTCACCGCTCTTCATG -3′ and GAPDH (GenBank: NM_008084): 5′- CATCACTGCCACCCAGAAGACTG -3′ and 5′- Reactions were performed in a 12.5 ll mixture containing 6.25 ll qPCRTM Green Master Kit for SYBR Green I (Jena Bioscience, Germany), 0.25 ll of each primer (200 nm.), three ll cDNA (100 ng), and 2.25 ll nuclease-free water. The PCR protocol used consisted of a 5-minute denaturation at 94 °C followed by 45 cycles of 94 ֯°C for 15 s and 60 ֯°C for 30 s. Reactions were performed in triplicate. Two separate reactions without cDNA or with RNA were performed in parallel as controls. Relative quantification was performed according to the comparative method and using Lightcycler 96 software. All qPCR analysis was performed according to the minimum information for publication of quantitative real-time PCR experiments (MIQE) guidelines [27].
Following RNA and DNA isolation from isolated tissues using the TriPure total RNA isolation kit per the manufacturer's instructions (Roche Molecular System, USA), total protein was precipitated, and its concentration was determined using the Bradford method. 25 ll of the Laemmli sample buffer supplemented with 2-mercaptoethanol at a final concentration of 7.5% (vol/vol) were combined with 25 ll of each protein sample (1 lg/ll). The samples were heated for 15 minutes at 65 degrees Celsius, separated using 10% SDS-PAGE, and then electrophoretically transferred to a nitrocellulose membrane (Schleicher & Schuell, Inc., Keene, NH). After being incubated for 1 hour in PBS containing 5% nonfat milk, the filters were blocked. Following a PBS-Tween wash, the blots were immunoblotted at a 1:500 ratio using a primary antibody against rat Bmal1 (Sanra Cruz Biotechnology Inc. sc-373955). Goat anti-rabbit HRP-conjugated antibody (Sanra Cruz Biotechnology Inc. sc-376938) and DAB reagent (Sigma Aldrich, Germany) were used to detect the primary antibody. Densitometric quantification of Bmal1 proteins in relation to GAPDH as a calibrator was performed using Image J software (National Institutes of Health). Western blot was done in three independent experiments for each sample.
Data analyses were done using the SPSS 26 software package (SPSS Inc., Chicago, IL, USA). One-way ANOVA was used to test differences between various means (post hoc analysis LSD test). All experimental data were presented as the mean ± SEM. The level of significance for all tests was set at P<0.05.
Results. The variance analysis of the study's findings is shown in Table 1. The results show that aerobic exercise affects the target parameters in diabetic mice at a 1% significance level. Following the intervention, Table 2 shows the average comparison of the assessed variables across all research groups. The differences between the columns in the LSD post hoc test are denoted by unequal Latin letters above them. The abbreviations CD-ZT3 stand for "light phase diabetic control group," CD-ZT15 for "dark phase diabetic control group," and TD-ZT3 for "light phase diabetic exercise group," respectively. CH-ZT3 stands for "light phase healthy control group," CH-ZT15 for "dark phase healthy control group," and CD-ZT3 for "light phase diabetic control group." The acronyms for HOMA-IR are insulin resistance index, oxidant to total antioxidant ratio, and Bmal1.
The post hoc test between research groups revealed that diabetic rats had significantly higher levels of glucose, insulin, HOMA-IR, and weight variables than healthy control rats (Table 2). Aerobic exercise had a significant impact on the training groups' changes in these variables' levels in comparison to the diabetic groups, as well as the trained rats' decreasing trend in these variables' levels. Changes in the levels of glucose, insulin, and HOMA-IR variables in all the study groups showed a significant difference in response to the two phases of darkness and light. Of course, there was no appreciable difference in weight changes between the two phases of darkness and light (P>0.05).
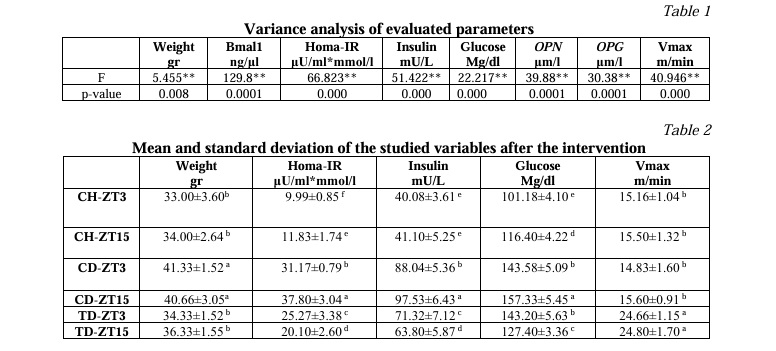
A comparison of the OPN variable's mean results (Fig. 1) revealed that rats with diabetes had significantly higher levels of this variable than rats in the healthy control group (P<0.05). The evaluation of the impact of aerobic exercise in the training groups compared to the diabetic groups without exercise revealed a trend of decreasing and significant OPN changes in trained rats at the 5% level (P<0.05). The OPN variable did not significantly differ between the diabetic and control training groups, as it changed during the two phases of darkness and light (P>0.05).
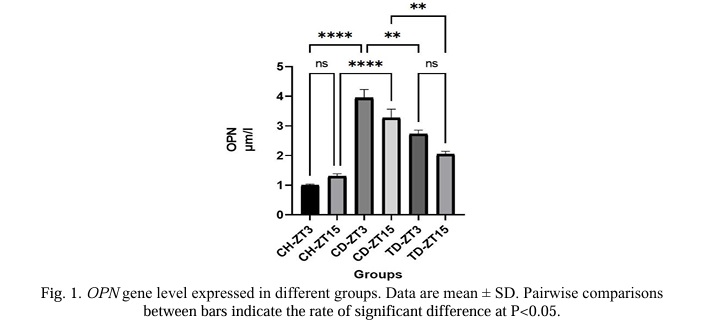
According to a comparison of the OPG variable's average results (Fig. 2), rats with diabetes had significantly higher levels of this variable in the light phase (P<0.05) than rats in the healthy control group, but these changes were not significant in the dark phase (P>0.05). The trend of OPG changes in the trained rats was decreasing and significant only in the light phase (P<0.05) when comparing the training groups' effects on diabetes groups without training to the diabetic groups. In the diabetic and control exercise groups, there were no appreciable differences in the changes in the two phases of darkness and light (P>0.05).
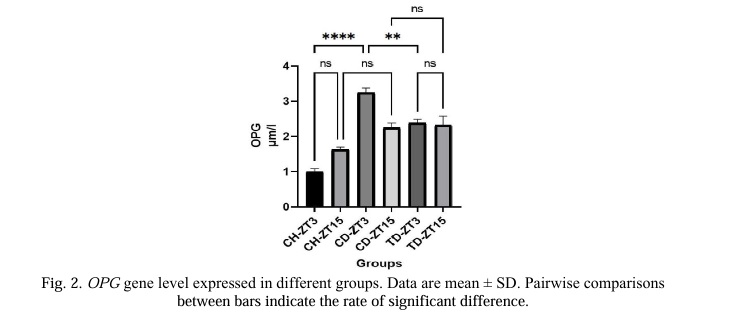
When the average results of the Bmal1 variable were compared (Fig. 3), it was evident that diabetic rats had significantly lower levels of this variable than healthy control rats (P<0.05). The comparison of the training groups' responses to aerobic exercise with the diabetic groups' responses revealed that the trained rats' response to this variable is increasing and that this trend is significant at the 5% level (P<0.05). Significant Bmal1 changes were observed in both the diabetic and control training groups (P<0.05).
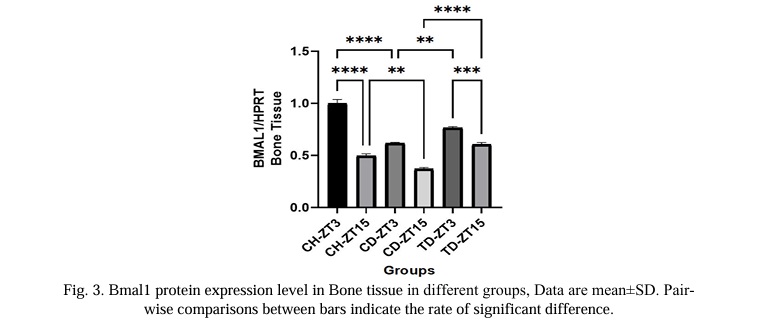
When comparing the maximum speeds of the various study groups, it was found that aerobic training significantly increased the maximum speed in the trained mice when compared to the untrained mice (P<0.05). Maximum speed variations during the two phases of darkness and light were not statistically significant (P>0.05).
Discussion. The results of this study showed that diabetes increased insulin resistance in diabetic rats and that eight weeks of endurance exercise significantly reduced this resistance in these subjects.Both glucose uptake and the activity of proteins involved in insulin signaling are enhanced by exercise [12]. In many studies [28, 29], improving insulin resistance and raising the insulin sensitivity of glucose-consuming cells. It has been verified that the findings largely agree with those of the current study. The present study's findings that endurance training reduced insulin resistance are consistent with those of Dela et al. [30], Park et al. [31], and other studies. This improvement in the circumstances results from increased insulin secretion and glucose uptake by consuming cells, according to Slentz et al. [29] and Malin et al. [32]. It should be noted that exercise improves beta cells' capacity to process signals and secrete insulin since disturbances in the behavior of lipids and intracellular pathways, including cellular stress responses like oxidative stress, endoplasmic reticulum stress, autophagy, and ceramide/LD formation, are involved in the death of beta cells caused by cytotoxicity [33].
The results of this study's analysis of changes in the OPN and OPG genes revealed that while endurance training causes a decrease in these genes in bone tissue, consumption of high-fat food causes an increase in these genes. However, there were no appreciable differences between the diabetic and control exercise groups in the changes in the two phases of darkness and light for the OPG and OPN variablesAccording to researchers, the type of exercise, its intensity and duration, and the recovery period between efforts all influence the OPG response [34]. In another study, in line with our findings, Humphries et al. (2009) investigated the effects of whole-body vibration with and without resistance training on bones for 16 weeks and concluded that bone vibration training and vibration with resistance training respectively led to a reduction of 50% and 36% in the amount of OPN [35].
According to Yu et al (2013), eight weeks of aerobic exercise running on a treadmill resulted in a reduction in serum OPN levels and fat percentage in young obese women, leading to the conclusion that changes in body composition are likely one of the factors that lowered OPN in the exercise group [12]. While having no impact on the phosphorylation pathway of protein kinase B (PKB) and mitogen-activated protein kinase (MAPK), OPN and OPG cause the proliferation of JNK and kinases regulated by extracellular messages (ERK) and impair insulin sensitivity by impairing PPAR-gamma and adiponectin gene expression. Therefore, it appears that by increasing insulin sensitivity and lowering inflammatory and pro-inflammatory factors, aerobic exercise can lower the expression of OPN and OPG levels [36].
According to the findings of the current study, eating foods high in fat causes the Bmal1 gene in bone tissue to decrease, which is consistent with the assertions made by Lee et al. [19] and Petrenko et al. [37]. In addition, the circadian clock is hampered in humans and cells in T2D, as evidenced by the reduction of Bmal1. A circadian rhythm may contribute to metabolic diseases, such as obesity and type 2 diabetes (T2D), reducing the exocytosis of insulin [38]. The present study shows that endurance exercise enhances Bmal1 status and increases Bmal1 levels in diabetic rats' bone tissue. Su and endurance exercise, regardless of the time, boost the amount of Bmal1, the primary protein that makes up the clock, even though HFD consumption, a factor that interferes with the circadian rhythm, decreases insulin-stimulated glucose uptake, while endurance exercise counteracts this effect [17].
Our study's findings show that there is a significant difference between the increase of the molecular clock gene Bmal1 during the dark phase and the light phase. Our results are in agreement with those of Savikj et al.[39], Dalbram et al.[17], Heden and Kanaley [40], and Mancilla et al. [41] among the studies conducted. Regarding the time of exercise in the present study, a significant difference was observed between the activity in the light phase and the dark phase of diabetic rats. Studies have found that participating in sports helps with circadian disorders because it can reset or change the phase of the body's internal clock. In fact, exercising at a specific time of day can correct circadian clock irregularities and improve energy metabolism [17]. Contrary to healthy individuals, those with T2DM have better circadian rhythms, insulin sensitivity, and glycemia at night. However, the conditions for activity are less favorable at night and in the early morning, which causes blood sugar and fat levels to rise in the morning. Found that melatonin reduces reactive oxygen species (ROS) and reactive nitrogen species (RNS) and acts as an antioxidant cascade [39]. Melatonin secretion in the dark phase increases with the reduction of light. This not only lessens the oxidative stress brought on by exercise but it also boosts the activity of antioxidant enzymes like superoxide dismutase. Melatonin secretion is influenced by physical activity, duration of activity, and time of day, either immediately or later. Melatonin and aerobic exercise both reduce the number of free radicals produced during exercise. Melatonin can lessen malondialdehyde and lipid peroxidation, particularly when combined with aerobic exercise. Additionally, it can improve the body's health and adaptation to physical activity by reducing ROS caused by that activity [42]. Additionally, diabetes results in a decrease in Bmal1, which affects both the body's ability to tolerate glucose and the ability of skeletal muscles to absorb glucose when stimulated by insulin. Contrarily, in mice with type 2 diabetes, upregulating Bmal1 expression enhances insulin sensitivity and enhances insulin tolerance [17]. Sympathetic activation and glucocorticoid release are likely to be the common signalling pathways by which physical activity alters the body's internal clock [25]. The oxidation of the NADPH cofactor and the displacement of the -subunit of the K+ channel, which ultimately results in the induction of sleep, are probably caused by the buildup of ROS. According to recent research, clock genes have redox characteristics and may be directly impacted by the molecular mechanism of adenosine accumulation. It works for intracellular interactions [24]. In this way, the prevention and management of type 2 diabetes may benefit from therapeutic approaches intended to improve circadian clock function. However, these findings did not agree with those of Steidle-Kloc et al. [44], or Chiang et al. [45]. In the study conducted by Kloc et al., the subjects were CAD/T2DM patients who voluntarily underwent a number of exercise sessions at home, reducing the amount of control over the proper execution of the exercise protocol. Acute resistance training that took place at 7 AM and 5 PM was the activity that Toghi-Eshghi and Yardley studied. The 12-week study by Chiang et al. included diabetics as well. The type and intensity of the activity, as well as the time spent engaging in the activity, can generally be mentioned as contributing factors to the studies' inconsistent findings. According to some research, Bmal1 is phosphorylated by insulin-activated PI3KAKT signaling, which lowers its transcriptional activity. In addition, because prolonged starvation can increase insulin signalling, refeeding after a prolonged fast has a stronger effect on the clock genes [16]. Because some strains of mice have higher levels of insulin resistance brought on by HFD, the type of subjects and mice used in the study can also affect the outcome [17].
Endurance exercise at the start of the dark phase increases the main clock protein, improves insulin resistance and bone index genes in type 2 diabetic rats. Limitations include uncontrolled food consumption, limited time measurements, and lack of evaluation of other circadian rhythm genes. Consideration of impacts on liver and muscle is important. Additionally, HIIT has been found in some studies to be more effective on cell metabolism than endurance exercises [17], and there was no room to compare them in the current study, so further research is necessary.
levels, potentially reducing the risk of bone tissue damage in diabetic patients. Limitations include uncontrolled food consumption, limited time measurements, and the need to consider the impact on other tissues like the liver and muscle.OPG and OPN genes in bone tissue. Daily use of such exercise can modify OPG and OPN, and Bmal1 protein levels in bone tissue. Performing eight weeks of endurance exercise at the beginning of the dark phase yields greater improvements in insulin resistance and Bmal1 protein concentration compared to the light phase. These findings can guide the development of exercise protocols aligned with the circadian rhythm to mitigate bone tissue damage in diabetic individuals. Regardless of the timing, endurance training for eight weeks reduces the expression of OPG, OPNThe study reveals that diabetes increases insulin resistance, Conclusion.





















Список литературы
Список использованной литературы появится позже.