Genetic variation of ERN1 and susceptibility to type 2 diabetes
Abstract
Background: The endoribonuclease IRE1 (ERN1) is an important sensor for the endoplasmic reticulum unfolded protein response (UPR), and its activation happens as a result of the accumulation of unfolded and misfolded proteins in the endoplasmic reticulum (ER). The ERN1 gene may be involved in ER stress, a feature of type 2 diabetes (T2D). The aim of the study: To investigate the relationship between common single nucleotide polymorphisms (SNPs) of the ERN1 gene and T2D risk. Materials and methods: The study included 1558 T2D patients (586 males and 972 females) and 1611 (618 males and 993 females) healthy subjects. Two common SNPs, such as rs196914 and rs9911085 located in the regulatory region of the ERN1 gene, were genotyped by the MassArray Analyzer-4 system. Results: Genotypes T/C-C/C rs196914 showed an association with an increased risk of T2D (OR=1.18, 95% CI 1.03-1.36, p=0.017). An associative analysis stratified by sex and BMI revealed that this association occurred in females with a BMI greater than 25 kg/m2 (OR = 1.20, 95% CI 1.03-1.40, p = 0.02). Furthermore, the genotypes rs9911085 T/C-C/C were linked to T2D risk in females with a BMI greater than 30 kg/m2 (OR = 1.44, 95% CI 1.02-1.95, p = 0.034). The haplotype rs196914C-rs9911085C was found to be associated (p=0.004) with T2D risk in overweight and obese subjects (i.e. BMI≥25 kg/m2). Conclusion: The present study was the first to show the impact of ERN1 gene polymorphisms on susceptibility to type 2 diabetes; however, the association was female- and BMI-specific. Further studies are required to confirm the association between ERN1 and T2D risk.
Keywords: type 2 diabetes mellitus, overweight, obesity, endoplasmic reticulum stress, misfolding, unfolded protein response, IRE1 pathway, ERN1
Introduction. Diabetes mellitus is one of the global health problems of the 21st century [1]. According to the International Diabetes Federation (IDF), diabetes mellitus will affect 537 million persons aged 20–79 years (prevalence 10.5 %) in 2021, and 783 million (prevalence 12.2 %) by 2045 [1]. At the same time, Russia ranks second among all European countries in terms of diabetes patients, with 90% of patients suffering from type 2 diabetes (T2D) [1].
Type 2 diabetes is a complex condition that involves both hereditary and environmental variables [2]. The pathophysiology of T2D is linked to pancreatic cell dysfunction and insulin resistance in peripheral tissues, however the core cause of these illnesses is unknown. The dysfunction of pancreatic beta-cells in T2D is primary and is detected at the earliest stages of the disease, and the progressive loss of beta-cells due to their apoptosis forms the basis for the progression of the disease and the development of complications [3, 4, 5].
It has been established that oxidative stress (OS), caused by a shift in redox homeostasis towards excessive formation of reactive oxygen and nitrogen species, plays a significant role in the development of dysfunction and apoptosis of beta-cells in T2D, and due to their low antioxidant capacity, beta-cells are extremely sensitive to oxidative damage [6, 7]. Endogenous glutathione deficit, which is pathogenetically related with beta cell malfunction and death, has been established in a number of studies to be the primary disturbance of redox homeostasis and OS formation in diabetes [8, 9, 10]. Many studies [11, 12, 13] show that chronic endoplasmic reticulum stress (ER) is involved in the creation of -cell dysfunction and death in T2D, in addition to oxidative stress.
It is well known that the synthesis and secretion of insulin in beta-cells is related to the ability of the ER to package, process, and move correctly folded insulin protein into the Golgi apparatus and then to the membrane surface for subsequent secretion [12, 14]. The accumulation of misfolded protein chains is the pathophysiological core of ER stress, a general biological phenomenon of functional overload of the protein secretion apparatus. When the maturation of protein molecules is disrupted, the cell loses its ability to function normally and faces death. Eukaryotes have evolved a complicated homeostatic mechanism known as the unfolded protein response (UPR) to restore proteostasis. This mechanism is represented by a triad of proteins – ERN1, PERK, and ATF6, each of which has a regulatory domain embedded in the ER lumen. Under normal physiological conditions, this domain is bound by the binding immunoglobulin protein (BiP) chaperone. The content of free chaperones in the ER lumen naturally decreases as the load on the apparatus grows, and BiP is released to perform its principal job of protecting protein maturation. ERN1, PERK, and ATF6 are now anchored in stress signaling pathways after being released from their relationship with BiP. As a result, the ER has a set of sensors that monitor intraluminal chaperone availability [15]. ERN1 has been identified as one of the key components of the UPR and a key regulator of gene expression. The cytoplasmic C-terminal region of ERN1 divides into two domains, each containing kinase and endoribonuclease activity. Dimerization happens after BiP separation, arranging kinase domains "face to face" [16]. The autophosphorylation in this case changes the structure of ERN1, opening the nuclease centers of its dimers and arranging them "back to back" [17]. As a result, ERN1 inhibits protein translation in the cytoplasm by selectively degrading a number of mRNAs. The degradation of CD59 mRNA in HeLa cells [18] and mRNA of the microsomal triglyceride transporter factor MTP in the intestinal epithelium, both of which impede chylomicron production [19], are two examples. There is also evidence of ERN1-mediated proinsulin mRNA degradation in chronic hyperglycemia [20].
Furthermore, from the group of genes responsible for UPR, only ERN1 is highly expressed in the pancreas and is considered a central regulator of ER stress signaling, playing a crucial role in the regulation of protein and insulin biosynthesis, in particular [21]. In their investigation, K. Haze and co-authors discovered that inhibiting ERN1 signaling in vitro lowers insulin production at the translation and protein folding levels [22]. When ERN1 is activated, it cleaves the mRNA for X-box binding protein 1 (XBP1), which activates the transcription factor for XBP1. Activated XBP1 starts various transcriptional processes that boost ER growth and biogenesis and increase protein entrance into the ER for maturation and/or cleavage of misfolded proteins after translocation to the nucleus. XBP1-deficient animals have poor -cell proliferation, proinsulin processing, and insulin production, as well as ERN1 hyperactivation, according to Lee et al. [23]. As a result, ERN1 is essential for optimum insulin production and ER homeostasis. There is no evidence in the literature that polymorphic variants of the ERN1 gene are linked to the development of type 2 diabetes, which was the premise for this investigation.
The aim of the study. To investigate the relationship between polymorphic variants of the ERN1 gene and the likelihood of acquiring type 2 diabetes.
Materials and methods. An ethnically homogeneous sample of 3229 unrelated citizens of Central Russia (mostly natives of the Kursk region) of Slavic nationality (self-identification) was used in the study. The KSMU Regional Ethics Committee authorized the study protocol (extract from protocol No. 10 dated 12.12.2016). The study included 1558 patients with T2D (586 men and 972 women) with a mean age of 61.1±6.9 years who were hospitalized at the endocrinology department of the Kursk City Clinical Emergency Hospital from November 2016 to October 2019. The diagnosis of T2D was made using WHO criteria [24, 25]. The control group consisted of 1611 apparently healthy volunteers, 618 men and 993 women with an average age of 60.8±5.7 years, who had previously donated blood to the Regional Blood Transfusion Station [26]. Based on written informed consent, all study participants were given 5 mL of fasting venous blood into vacuum tubes Vacuette with EDTA as an anticoagulant. The genomic DNA was extracted using phenol-chloroform. Two single nucleotide polymorphisms in the ERN1 gene, rs196914 (T>C) and rs9911085 (T>C), were chosen for molecular genetic investigation because they had a strong regulatory potential and a minor allele frequency of at least 10%. The MassArray Analyzer 4 platform was used to detect ERN1 gene polymorphisms using laser desorption/ionization time-of-flight mass spectrometry (Agena Bioscience). The SNPStats web program (https://www.snpstats.net/start.htm) was used to statistically process the collected data. Co-dominant, dominant, recessive, over-dominant, and log-additive genetic models were investigated. The association was considered significant at P<0.05. KEGG pathways tools were used to recreate metabolic pathways, and eQTLGen (www.eqtlgen.org) was used to investigate tissue-specific expression of the ERN1 gene.
Results and discussion. Hardy-Weinberg equilibrium was achieved for ERN1 genotype frequencies. Table 1 presents the results of the analysis of associations with the risk of developing type 2 diabetes in the general sample, as well as separately in men and women. In the whole sample, T/C-C/C rs196914 genotypes were associated with an increased risk of acquiring the disease (OR=1.18, 95% CI 1.03-1.36, p=0.02, dominant genotypic model). The rs9911085 ERN1 gene was not linked to the likelihood of acquiring T2D.
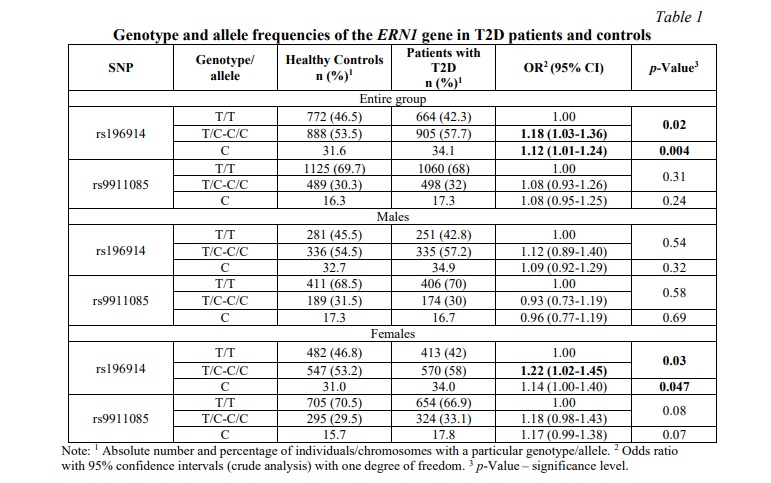
Separate comparison of male and female patients with T2D with controls showed that the T/C-C/C rs196914 association of the ERN1 gene was typical only for women (OR=1.22, 95% CI 1.02-1.45, p=0.03, dominant model), while in in men, an increased risk of the disease with this genotype was not observed (p>0.05).
Following that, a stratified analysis by sex and BMI was conducted (Table 2): the association found in the general group was observed only in women with BMI ≥25 kg/m2, which corresponds to overweight and obesity (OR=1.20, 95% CI 1.03- 1.40, p=0.02, dominant model) and was absent in men (p>0.05). In addition, an association of the rs9911085 T/C-C/C polymorphism in obese women (BMI≥30 kg/m2) was established: OR=1.44, 95% CI 1.02-1.95, p=0.03, dominant model.
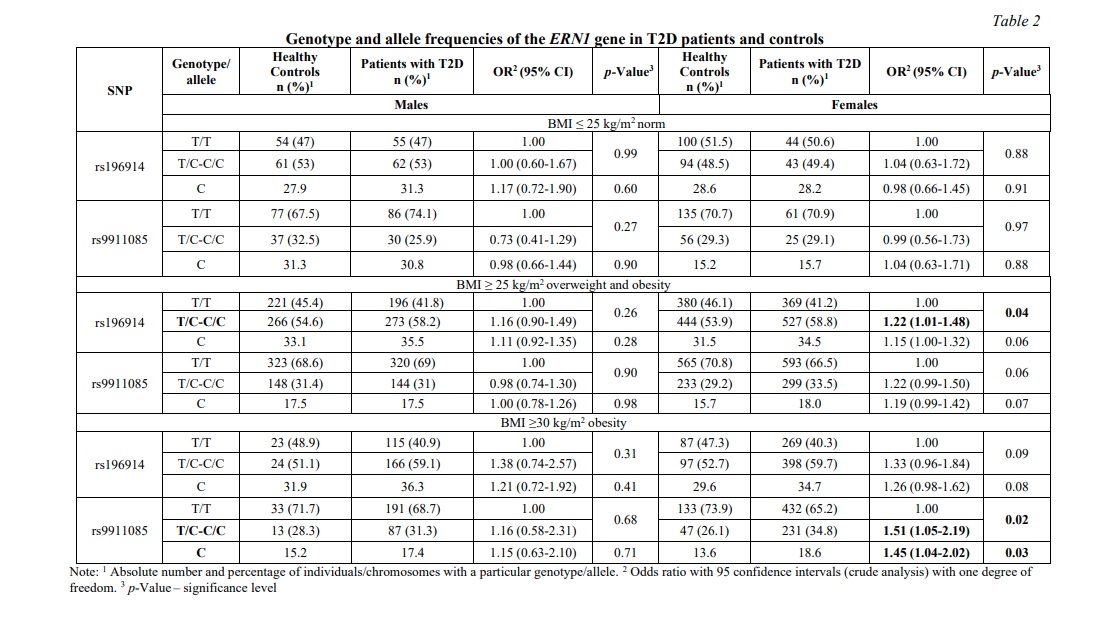
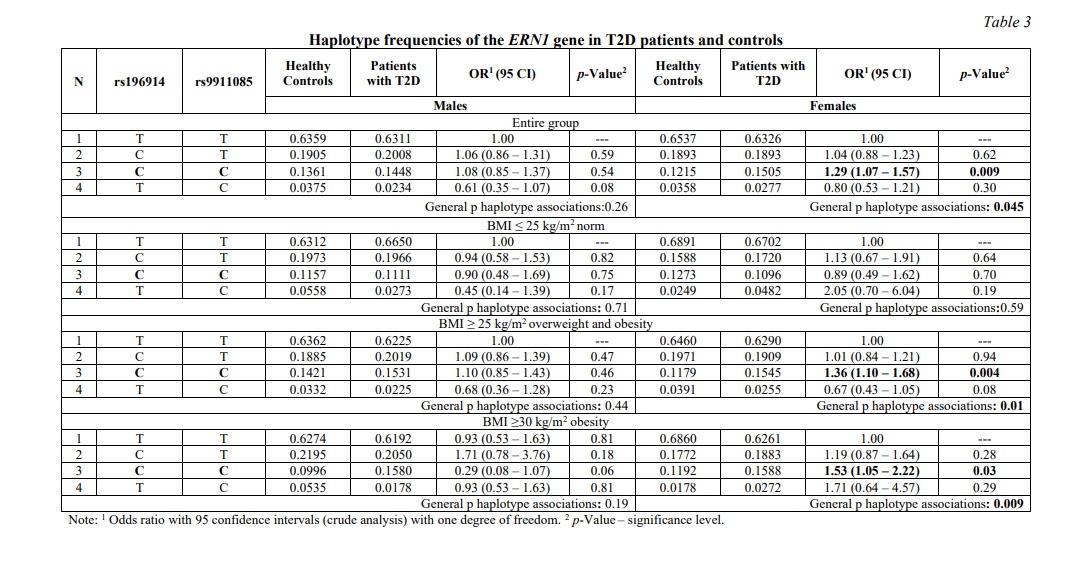
Table 3 shows the results of the distribution of haplotype frequencies in T2D patients and healthy people. Haplotype rs196914С-rs9911085С was associated with an increased risk of developing T2D both in women in the general sample and in women with overweight and obesity (BMI ≥25 kg/m2): OR=1.36, 95% CI 1.10-1.64, p=0.004 .
We used the eQTLGen consortium database to examine the mRNA expression profiles (eQTL) of the studied SNPs, which include data from a thorough transcriptome investigation of 30,847 blood samples from healthy volunteers, the majority of whom are of European origin. The database made it possible to identify significant cis-eQTLs for each studied SNP. Thus, the alleles rs196914-C and rs9911085-C are associated with a significant decrease in the expression of the ERN1 gene in whole blood (P=5.10×10-140 and P=5.17×10-138, respectively, FDR≤0.0001). The alternative alleles rs196914-C and rs9911085-C are related with hypermethylation of the ERN1 gene, and therefore with a decrease in its expression in childhood and adolescence, according to an analysis of the effects of the examined polymorphisms on gene methylation status (mQTL). According to experimental evidence from K.L. Lipson et al., suppressing ERN1 expression lowers the amount of cellular insulin [20].
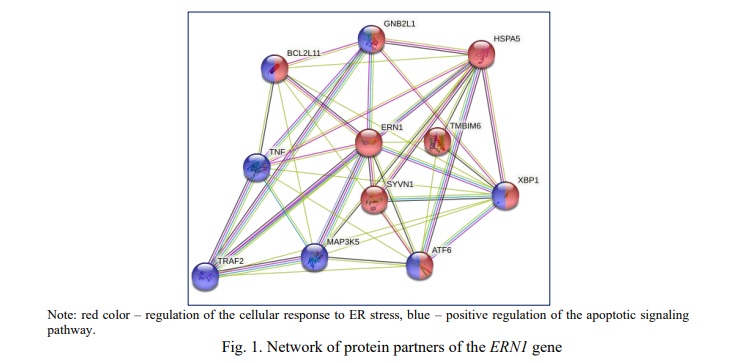
The STRING tool was used to analyze ERN1 connections with other genes at the level of protein products, and it revealed functional partners of ERN1 that constitute an interacting network of 10 proteins (Fig. 1): HSPA5 – belongs to the heat shock protein family 70, glucose-regulated Bip ER chaperone ; XBP1 – X-box-binding protein 1; ANF6 – activating transcription factor 6, initiates UPR during ER stress; TMBIM6 – Bax inhibitor, apoptosis suppressor; SYVN1 – E3 ubiquitin protein ligase synoviolin; GNB2L1 – activated C-kinase 1 receptor; BCL2L11 – Bcl-2-like protein 11, induces apoptosis; MAP3K5 – mitogen-activated protein kinase kinase 5; TNF- tumor necrosis factor; TRAF2 – TNF receptor-associated factor 2. Using the analysis of gene ontology terms enrichment, it was found that 8 proteins are directly involved in the regulation of the cellular response to endoplasmic reticulum stress: HSPA5, ANF6, XBP1, SYVN1, BCL2L11, TMBIM6, GNB2L1 (FDR=5.66*10-13); and 7 proteins are responsible for the positive regulation of apoptosis: ANF6, XBP1, BCL2L11, GNB2L1, MAP3K5, TNF, TRAF2 (FDR=4.86*10-6).
Glucotoxicity and lipotoxicity impede ER stress compensation in the established clinical picture of diabetes mellitus [27, 28]. ER stress plays a key role in the creation of obesity and insulin resistance in adipose and hepatic tissue, in addition to a decrease in insulin production due to a decrease in the population of β-cells. Signaling through the unfolded protein response's ERN1 pathway has both beneficial and harmful consequences in obesity. Overexpression of the ERN1-regulated transcription factor XBP1 protects cells from metabolic balance abnormalities caused by obesity [28]. However, ERN1 hyperactivation involves a second mechanism, the dependent breakdown and pro-inflammatory signaling of TRAF2/JNK. TRAF2 is a receptor that signals TNF ligation to turn on apoptosis via MAPK, JNK and p38 (Fig. 2).
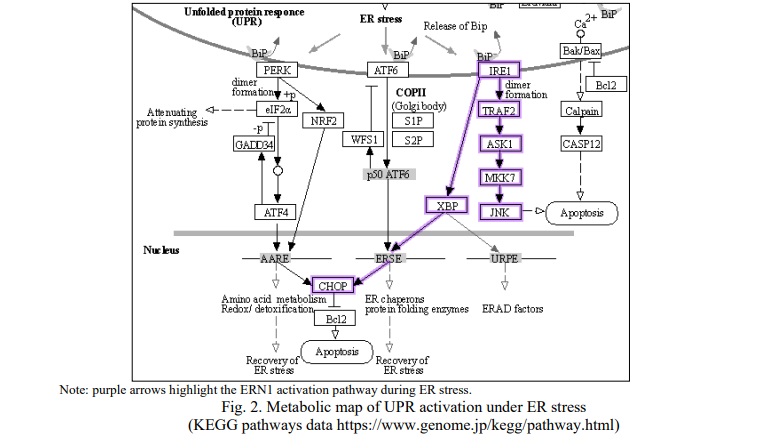
The carriage of the rs196914-C and rs9911085-C alleles is associated with a decrease in the expression of serine/threonine protein kinase/endoribonuclease (ERN1), which is a central regulator of ER stress signaling. Reduced ERN1 expression can result in signaling reduction of the XBP1 transcription factor, which causes chaperone activation to worsen (Fig. 2), lowering insulin production at the translation and protein folding levels. At the same time, overweight and obesity are two factors that impede protein folding [29]: poorly folded proteins accumulate in the cell due to a decrease in ERN1 activity, which can lead to the cell's forced death.
Conclusion. Thus, the study was the first to show a link between the polymorphic loci rs196914 and rs9911085 in the regulatory area of the ERN1 gene with an elevated risk of developing T2D, indicating that the endoribonuclease IRE1 gene may play a role in the disease etiology. The discovered connections are unique to women and are influenced by body mass index. The decreased transcriptional activity of the ERN1 gene in carriers the of alternative alleles rs196914-C and rs9911085-C of the analyzed polymorphisms can explain the link between the studied loci and illness. To confirm connections between polymorphic variants of the ERN1 gene and the likelihood of acquiring type 2 diabetes, more research in various communities around the world is needed.
Financial support
The study was supported by the Russian Science Foundation (project №22-25-00585).
Thanks
The author expresses her gratitude to Professor Alexey Polonikov and Associate Professor Iuliia Azarova from the Research Institute for Genetic and Molecular Epidemiology (Kursk State Medical University) for their in-depth assistance in this research





















Reference lists