Mitigation of indomethacin-induced gastric inflammation in mice through the administration of ethanol extract from Hedyotis capitellata Wall. leaves
Abstract
Background: Gastritis, a prevalent global malady, stems from various factors such as unhealthy lifestyles, bacterial infections, or non-steroidal anti-inflammatory drug usage. If untreated, it leads to severe complications. Hedyotis capitellata Wall., recognized for its medicinal properties, is traditionally acknowledged for its effectiveness in pain relief, neutralizing gastric acid, reducing acid reflux, and promoting ulcer healing. The aim of the study:This study delves into exploring the preventive and protective prowess of H. capitellata leaf ethanol extract (EEHC) against indomethacin (IND)-induced gastric mucosal damage in mice. Materials and methods: EEHC administered at 300, 350, and 400 mg/kg doses, with omeprazole (OME) (20 mg/kg) as the reference standard, EEHC's impact was assessed on gastric acid levels, mucosal damage, malondialdehyde (MDA) content, antioxidant activity (catalase, myeloperoxidase, glutathione, total antioxidant capacity), cytokine levels (TNF-α, IL-1β, IL-6, IL-10), and histopathology. Results: EEHC, particularly at 400 mg/kg, exhibited maximal inhibition of total gastric acid (8.58 ± 0.16 mEq/l), decreased MDA content (5.29±0.15 nM/g tissue), and lowered pro-inflammatory cytokines (TNF-α: 224.04±17.56 pg/ml) compared to IND-treated mice (p<0.05). Dose-dependent EEHC enhanced the antioxidant defense system by reducing catalase and myeloperoxidase activity and increasing glutathione and total antioxidant capacity levels (p<0.05). Mice treated with EEHC displayed significantly lower gastric ulcer indices across all experimental models compared to the IND-treated group (p<0.05). Histopathological examinations further affirmed EEHC's anti-inflammatory efficacy. Conclusion: The study establishes that oral administration of EEHC exerts a positive protective influence on the gastric mucosa, augmenting the antioxidant defense system and anti-inflammatory effects. H. capitellata emerges as a promising resource for managing gastric inflammation due to its potent antioxidant properties
Introduction. Gastric inflammation, known as gastritis, is a prevalent global disease affecting approximately 10% of the world’s population. It represents a significant health issue that greatly impacts the quality of life for millions of individuals [1]. The pathophysiology of this digestive disorder is characterized by an imbalance between protective factors of the gastric mucosa, such as bicarbonate, prostaglandins, peptides, growth factors, gastric mucus, and the innate immune response of mucosal cells, and aggressive factors including acid, pepsin, H. pylori, non-steroidal anti-inflammatory drugs, tobacco, alcohol, trauma, shock, and stress. The progression of this condition is influenced by the dominance of disruptive factors over defensive mechanisms [2]. Gastric acid secretion is primarily regulated by histamine 2 receptors (H2r) binding to histamine, stimulating acid secretion into the stomach, which is subsequently accelerated by the cAMP/protein kinase A/proton pump pathway. Therefore, most therapeutic approaches for gastric inflammation involve the use of histamine 2 receptor antagonists (H2RAs) such as ranitidine and cimetidine, as well as proton pump inhibitors (PPIs) like pantoprazole and lansoprazole. Although these medications have demonstrated clinically acceptable efficacy, adverse effects such as rash, itching, constipation, nausea, abdominal pain, and diarrhea have been reported [3]. Due to the potential side effects associated with synthetic drugs, the development of natural products for the treatment of gastric inflammation, which offer minimal adverse effects, is highly valued. Recent studies in the field of gastrointestinal disorders have focused on exploring the potential role of natural remedies, utilizing readily available plant-based medicines as primary sources for the development of new drugs due to their safety, minimal side effects, affordability, effectiveness, and relatively low toxicity [4].
Hedyotis is a flowering plant genus in the Rubiaceae family. It is widely distributed in tropical and subtropical regions, comprising approximately 500 species. Previous studies have revealed the chemical diversity of Hedyotis plants, including secondary metabolite groups such as alkaloids, anthraquinones, flavonoids, lignans, iridoids, and triterpenoids. This chemical diversity has provided various important pharmacological activities, including antibacterial, anti-diabetic, antioxidant, anti-inflammatory, and anticancer effects [5]. Extracts from Hedyotis species such as H. capitellata, H. dichotoma, H. verticillata, H. herbacea, H. pinifolia, H. corymbosa, and H. nudicaulis have demonstrated antioxidant, free radical scavenging, anti-inflammatory, cytotoxic, and antibacterial properties [6]. H. diffusa (a species of Hedyotis) is commonly used in folk medicine to treat lung heat, asthma, cough, gastric inflammation, and improve colonic mucosal damage [7]. H. corymbosa, a traditional medicinal plant belonging to the Hedyotis genus, contains compounds such as alkaloids, anthraquinones, lignans, triterpenes, flavonoids, and glycosides. It exhibits anti-inflammatory, anticancer, hepatoprotective, neuroprotective, gastric mucosal protective, and cytotoxic properties [8]. In Vietnam, Hedyotis capitellata Wall. (a species of Hedyotis) is a traditional remedy used for diuretic, detoxification, and anti-inflammatory purposes for the stomach [5]. H. capitellata contains alkaloids, saponins, tannins, and anthraquinones. Its extract has been demonstrated to possess effective antioxidant, anti-inflammatory, and antibacterial activities [9]. In traditional Vietnamese medicine, H. capitellata is utilized to alleviate pain, neutralize gastric acid, reduce acid reflux, and heal ulcers [10]. However, the broad claims about the health benefits of H. capitellata primarily rely on observations and anecdotal evidence rather than scientific substantiation. Experimental evidence supporting the anti-inflammatory effects of H. capitellata on gastric inflammation is still lacking. Therefore, the objective of our study is to evaluate the anti-inflammatory activity of the ethanolic extract of H. capitellata leaves by using an indomethacin-induced gastric inflammation mouse model and assess the effects of this extract on oxidative stress parameters in the mouse stomach tissue.
The aim of the study. To assess the efficacy of H. capitellata leaf ethanol extract (EEHC) in alleviating gastric mucosal damage.
Materials and Methods
Drugs and chemicals
Indomethacin (IND) and Omeprazole (OME) were purchased from Pharmedic Pharmaceutical Company, Vietnam. All other chemicals were analytical grade and obtained from Sigma (St. Louis, MO, USA).
Plant material and preparation of the extract
Collection of plant material: The leaves of H. capitellata, belonging to the genus Hedyotis and the family Rubiaceae, were collected in Cẩm Mỹ district, Đồng Nai province, Vietnam in March 2023. The plant specimen with the number HC140323VST was deposited at the Plant Biotechnology Laboratory, Institute of Biotechnology and Food Technology, Ho Chi Minh City University of Industry, Vietnam.
Preparation of the extract: The leaves of H. capitellata (depicted in Fig. 1A) underwent a meticulous cleansing process, followed by air-drying in shaded conditions. Subsequently, the German-manufactured Memmert UN110 drying oven was employed to desiccate the leaves of H. capitellata at 60°C until achieving a stable weight. The dry leaves, possessing a moisture content of 5%, underwent pulverization (180/125) using a medicinal grinding machine (TMND-A01, Tan Minh Co. Ltd., Vietnam), and were after that conserved in a refrigerator. Approximately 250 g of the leaf powder was immersed in 800 ml of 96% ethanol for 72 hours. Following this maceration period, the mixture underwent filtration utilizing Whatman No. 4 filter paper, with the resultant upper layer being concentrated through a rotary evaporator (IKA, Werke GmbH & Co. KG, Germany) at 60°C. The resultant concentrated extract, denoted as EEHC (Fig. 1B), was then stored at 4°C pending further analytical procedures.

Phytochemical screening and determination of total phenolic (TPC), total flavonoid (TFC) content of extract
Screening of phytochemicals: The ethanol extract derived from H. capitellata leaves underwent a screening process to detect the presence of alkaloids, tannins, steroids, and saponins, as outlined by Muhamad et al. [11]. Qualitative assessments were conducted based on the outcomes observed in each experimental procedure.
For the alkaloid test, approximately 1 ml of crude extract (at a concentration of 10 mg/ml) was combined with 160 μl of Dragendorff's reagent, consisting of 12 ml of 1.33 M bismuth nitrate in 30% nitric acid and 50 ml of 3.26 M potassium iodide adjusted to 100 ml with distilled water. The identification of alkaloids was benchmarked against the standard compound, caffeine (10 mg/ml). The steroid screening utilized the Salkowski test, wherein 1 ml of crude extract (10 mg/ml) was thoroughly mixed with 500 μl of chloroform. Subsequently, 1 ml of concentrated sulfuric acid was added to the mixture. The presence of steroids was compared to the standard compound, cholesterol.
The tannin test was determined by introducing 90 μl of 1% w/v ferric chloride solution to 10 mg of crude extract dissolved in 1 ml of methanol. The presence of tannins was then compared to the standard compound, gallic acid. In addition, the determination of saponin content involved mixing 90 μl of dimethylsulfoxide (DMSO) and 5 ml of distilled water with 10 mg of crude extract dissolved in 1 ml of ethanol. The resulting mixture was vigorously shaken, and the presence of saponins was compared to the standard compound, sodium dodecyl sulfate (SDS).
Determination of total phenolic content (TPC)
The assessment of phenolic compounds in the extract followed a spectrophotometric methodology as delineated by Mansoori et al., with slight modifications [12]. The preparation of the crude extract solution involved dissolving approximately 0.5 ml (1 mg/ml) of the crude extract in 0.5 ml of methanol. Subsequently, 100 µl of the resulting stock sample was introduced to 500 µl of Folin-Ciocalteau reagent, with thorough mixing for 1 minute. Following this, 2.5 ml of a 7.5% Na2CO3 solution was incorporated into the mixture, and the entire solution underwent stirring to achieve homogeneity of the dilute phase. Simultaneously, a blank sample was prepared similarly, utilizing 0.5 ml of methanol instead of the crude extract solution. After subjecting the samples to incubation at 45°C for 45 minutes in darkness within a thermostatic chamber, the absorbance was measured at 730 nm using a spectrophotometer. Three replicates were conducted to determine the average sample value. A gallic acid standard curve was established within the range of 20-100 μg/ml. Ultimately, the concentration of phenolic compounds was expressed in terms of gallic acid equivalents (mg GAE/g dw).
Determination of total flavonoid content (TFC): The determination of total flavonoid content (TFC) in the extract followed the AlCl3 colorimetric method, as outlined by Mansoori et al. with minor adjustments [12]. The extract solution was methanol-diluted to a concentration of 1 mg/ml, and a standard curve was established using quercetin dissolved in methanol within the range of 20-100 μg/ml. A solution comprising 2.0 ml of the diluted extract/quercetin, AlCl3 solution (0.1 ml of 10%, w/v), and CH3COOK (0.1 ml of 0.1 mM) was prepared, and the entire solution was made up of methanol. After incubating for 30 minutes at 25°C, the absorbance was measured at 415 nm. Ultimately, the total flavonoid content in the extract was quantified in terms of milligrams of quercetin equivalents per gram of the extract's dry weight (mg QE/g dw).
Experimental animals
Swiss albino mice with a weight range of 30-35 g were employed to assess the anti-inflammatory activity induced by indomethacin. These mice were sourced from the Pasteur Institute in Ho Chi Minh City, Vietnam. The animals were housed in standard laboratory conditions within glass cages, maintaining a 12-hour light/dark cycle, controlled temperature (24±2°C), and supplied with a commercial pellet diet designed for rodents, along with free access to water. To acclimate the mice to the environment, a 7-day adaptation period was observed before the commencement of the experiment. The handling and treatment of the animals adhered to the general guidelines for research methods and the evaluation of traditional medicine established by the World Health Organization in 2000 [13]. The experimental procedures followed the principles outlined in the Helsinki Declaration, emphasizing the protection of vulnerable research participants [14]. All interactions with experimental animals were carried out by personnel well-versed in the regulations for the use and care of laboratory animals. All experimental procedures were approved by the Ethics Committee for Animal Research of the University and adhered to the principles of care and use of laboratory animals. The use of laboratory animals is aligned with the National Guidelines on Ethics in Biomedical Research, as outlined by the Ministry of Health, Vietnam [15].
Experiment design
The experimental subjects were allocated into six groups, each consisting of five mice. The normal control group (DW group) received distilled water (10 mg/ml). The negative control group (IND group) was administered indomethacin (45 mg/kg) to induce gastric inflammation. The positive control group (IND+OME group) experienced gastric damage due to indomethacin and received omeprazole (20 mg/kg), a standard drug utilized in the therapy of gastric inflammation. The experimental groups, characterized by indomethacin-induced gastric lesions, were subjected to treatment with EEHC at concentrations of 300, 350, and 400 mg/kg (IND+EEHC300, IND+EEHC350, and IND+EEHC400 groups, respectively). The mice underwent a 24-hour fasting period and were orally administered indomethacin (45 mg/kg) to induce gastric inflammation. The standard drug and test compounds were administered orally 30 minutes before the administration of indomethacin. Forty-eight hours post-induction of gastric inflammation, the mice were treated once daily for 21 days with either the vehicle (distilled water, 10 ml/kg), the standard drug omeprazole (20 mg/kg), or EEHC at doses of 300, 350, and 400 mg/kg. On the subsequent day, blood samples were collected via cardiac puncture and stored at 4°C for subsequent analysis. Following this, the mice were anesthetized using a xylazine-ketamine anesthesia mixture [ketamine (80 mg/kg) and xylazine (10 mg/kg, IP)] and euthanized through CO2 gas inhalation. Surgery was conducted, and the stomach was dissected and opened along the greater curvature. After rinsing with sterile saline, the stomach was macroscopically evaluated for damage. The collected stomach samples were preserved at -80°C for further analysis.
Stomach acidity assessment
The determination of gastric acid levels followed the protocol outlined by Qureshi et al. [2]. The stomach was meticulously excised, maintaining closure at the lower end of the esophagus, and subsequently dissected open along the greater curvature to extract its contents. The obtained gastric fluid underwent collection, followed by centrifugation at 1000 rpm for 10 minutes. After centrifugation, the samples were decanted, and the volume of the gastric fluid was meticulously documented.
Gastric pH determination: A volume of 1 ml from the supernatant was diluted to 10 ml with distilled water. The pH of the resulting solution was measured using a digital pH meter (HI8424, Hanna Instruments Co. Ltd., USA).
Estimation of free and total acidity (TA): In a 100 ml conical flask, 1 ml of gastric juice was carefully pipetted, and 2 or 3 drops of Topfer reagent were introduced. The solution underwent titration with 0.01 N NaOH until the disappearance of all red color spots, resulted in a light yellow hue. The volume of alkali added during this phase was meticulously documented, representing the free acid content. Subsequently, 2 or 3 drops of phenolphthalein solution were incorporated, and the titration continued until a distinct pink color manifested. Once again, the total volume of alkali added was recorded (VNaOH), corresponding to the total acid content. The quantification of free and total acid was computed using the formula below:
 TA mEqL= VNaOH× CNaOH0.1 × 100mEqL
TA mEqL= VNaOH× CNaOH0.1 × 100mEqL  (Eq. 1)
(Eq. 1)
TA mEqL= VNaOH× CNaOH0.1 × 100mEqL  Evaluation of gastric mucosal damage (ulcer index and inhibition rate)
Evaluation of gastric mucosal damage (ulcer index and inhibition rate)
The stomachs of the experimental subjects were dissected along the greater curvature and scrutinized under a microscope by two investigators to identify lesions in the glandular region. A semi-quantitative scoring system, ranging from 0 to 4 (as detailed in Table 1), was established based on the severity of congestion and hemorrhage. This scoring system adhered to the methodology outlined by Tastekin et al. with slight modifications [16].
The gastric ulcer index (UI) and ulcer inhibition percentage (PUI %) were calculated according to the following formula:
UI= UI1+UI2+…+UIn n 
 (Eq. 2)
(Eq. 2)
where n is the total number of mice in each group
 (Eq. 3)
(Eq. 3)

Hematology and biochemistry assessment
On the final day of the experiment, blood samples were obtained from the animals through cardiac puncture and stored at 4°C until further analysis. An automated hematology analyzer, TC-Hemaxa 1000 (USA), was employed to quantify red blood cells (RBCs), white blood cells (WBCs), and various white blood cell types, including lymphocytes (LYM), monocytes (MONO), and granulocytes (GRA). The erythrocyte sedimentation rate (ESR) was determined using the Westergren pipette method. The serum, obtained after blood clotting, underwent centrifugation for 15 minutes at a speed of 3000 rpm. The levels of C-reactive protein (CRP) and alkaline phosphatase (ALP) were then analyzed utilizing an automated biochemical analyzer, TC-3300 Plus (USA).
Quantification of malondialdehyde (MDA)
The assessment of lipid peroxidation was conducted by evaluating the malondialdehyde (MDA) formation, employing the thiobarbituric acid method as detailed by Lim et al. with slight modifications [17]. The gastric mucosa layer was homogenized in a 10 ml solution of 100 g/l KCl. Subsequently, 500 µl of the homogenate was introduced into a new tube containing a thiobarbituric acid solution composed of 1.5 ml of 1 g/l 2-thiobarbituric acid, 0.2 ml of 80 g/l SDS, 1.5 ml of 200 g/l acetic acid, and 0.3 ml of distilled water. The mixture was heated at 98°C for 1 hour, allowed to cool to room temperature, and then combined with 5 ml of n-butanol: pyridine (15:1; Sigma-Aldrich). Following centrifugation at 3000 × g for 30 minutes, the absorbance of the upper layer was measured using a UV/Vis spectrophotometer at a wavelength of 532 nm. A standard curve was generated using 1,1,3,3-tetramethoxypropane (Sigma-Aldrich), and the results were expressed as nM MDA/g wet tissue (nM/g tissue).
Antioxidant assessment
Catalase (CAT) activity measurement: The determination of CAT activity in the tissue followed the protocol established by Lim et al. [17]. The gastric tissue underwent homogenization, with subsequent removal and washing of the glandular portions using cold physiological saline. Half of the gastric tissue was homogenized in a chilled phosphate-buffered saline solution (0.1 mol/l). The resulting homogenates were subjected to centrifugation at 4,500 rpm for 15 minutes at 4°C, yielding the supernatant representing the homogenized gastric tissue solution. The gastric mucosal homogenates were appropriately diluted with a buffer solution and then mixed with 1 ml of 30 mM H2O2. The absorbance was measured at a wavelength of 240 nm. CAT activity was expressed as mM per minute per milligram of tissue.
Measurements of mucosal myeloperoxydase (MPO) levels: The MPO content was quantified utilizing the assay method outlined by Araújo et al. with minor adjustments [1]. Gastric tissue samples were immersed in a Tris hydrochloric acid (HCl) buffer solution at a ratio of 1:5 (w/v), pH = 7.4, and minced with scissors for 15 seconds on a chilled ice plate. Homogenates were prepared in 0.5% hexadecyl trimethyl ammonium bromide (pH = 6.0), dianisidine dihydrochloride staining solution, potassium phosphate buffer solution (PBS), and 0.05% hydrogen peroxide (1%). Subsequently, the mixture underwent centrifugation (2000 × g for 20 minutes at 4°C). The absorbance of the samples was measured at a wavelength of 450 nm. The results were expressed as the quantity of enzyme capable of degrading 1 μmol/ml of peroxide at 25°C and reported as MPO units per gram of tissue (U/g tissue).
Estimation of glutathione (GSH) content: The determination of GSH content followed the method previously outlined by Sedlak and Lindsay with slight adjustments [18]. Gastric tissue was homogenized in 2 ml of Tris-HCl buffer solution [50 mM Tris HCl, 0.2 mM sucrose, and 20 mM EDTA; pH 7.5], precipitated with 0.1 ml of 25% trichloroacetic acid, and then subjected to centrifugation at 3500 × g for 40 minutes at 4°C to eliminate the precipitate. The GSH content in the supernatant was ascertained by measuring the absorbance at 412 nm, and the results were expressed as nM/mg of tissue.
Estimation of total antioxidant capacity (TAC): The concentration of TAC was determined using the method described by Popović et al. [19]. The reaction mixture contained 200 μl of reagent I (acetate buffer CH3COONa/CH3COOH, 0.4 M, pH 5.8), 20 μl of gastric tissue homogenate, and 20 μl of reagent II (10 mM ABTS, 2 mM H2O2 in acetate buffer (30 mM), pH 3.6). After 5 min, the absorbance was measured at 660 nm using a spectrophotometer, and the results were expressed as nM/mg protein.
Measurement of inflammatory markers and cytokines
The concentrations of cytokines TNF-α, IL-1β, IL-6, and IL-10 in gastric tissue were determined utilizing a mouse ELISA kit, following the methodology outlined by Danisman et al. [20]. The samples were incubated at 37°C for 90 minutes upon addition to the plates. After emptying the wells, the biotin-conjugated detection antibody working solution was introduced and incubated for an additional 60 minutes. After washing the plate, the horseradish peroxidase-conjugated working solution was added and incubated at 37°C for 30 minutes. Finally, the substrate solution and stop solution were sequentially added, and the absorbance was measured at 570 nm using a Multiskan™ GO Microplate spectrophotometer (USA).
Histopathology examination
After the evaluation period, samples of gastric tissue were gathered. The gastric wall tissue was preserved in a 10% formaldehyde solution for 48 hours and subsequently embedded in paraffin blocks. Sections with a thickness of 4 μm were obtained from each paraffin block and subjected to staining with hematoxylin and eosin (H&E) for histological assessment. The designated sections were scrutinized under a light microscope (Olympus BX 51, Japan) as per the protocol described by Danisman et al. [20].
Statistical analysis
The data were expressed as mean ± standard deviation and statistical comparisons were conducted using Statgraphics Centurion XIX (Statgraphics Technologies, Inc., USA) employing one-way analysis of variance (ANOVA), followed by Dunnett's post hoc test. Statistical significance was established when p-values were less than 0.05.
Results and Discussion
Phytochemical and quantification of total phenolic content (TPC) and total flavonoid content (TFC) in extract
The qualitative analysis of the chemical constituents of the ethanol extract from H. capitellata indicated the presence of tannins, flavonoids, steroids, alkaloids, saponins, and phenolics. The results, presented in Table 2, employed a notation system where a (+) symbol denoted the presence, while a (-) symbol denoted the absence of specific plant chemical compounds in H. capitellata. Quantitatively, the EEHC exhibited a total phenolic content of 67.85±4.29 mg GAE/g and a total flavonoid content of 41.64±3.48 mg QE/g, as outlined in Table 3. The results indicate that the EEHC contains various components that may be associated with anti-inflammatory and anti-oxidative activities. Among these components, the presence of tannins, flavonoids, and phenolics, known for their antibacterial properties and anti-inflammatory effects, suggests the potential application of the extract in treating gastric inflammation. Tannins may contribute to protecting the gastric mucosa from damage and exhibit anti-inflammatory effects. Flavonoids, as a type of compound with anti-oxidative and anti-inflammatory properties, could play a crucial role in reducing inflammation and safeguarding the gastric mucosa. Phenolic compounds are also recognized for their antibacterial and anti-inflammatory capabilities.
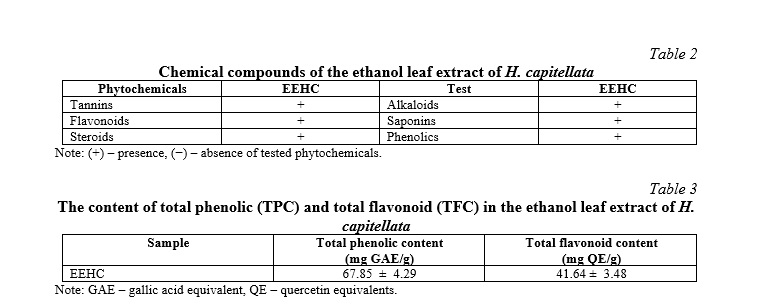
Plant chemical analysis of the ethanol extract of H. capitellata leaves revealed the presence of secondary metabolites previously reported in other medicinal plants (Table 2). To the best of our knowledge, this study is the first to report the presence of tannins, flavonoids, steroids, alkaloids, saponins, and phenolics in H. capitellata. Alkaloids are the end products of plant metabolism and serve as a nitrogen reserve, an essential nutrient for plant growth. These plant metabolites have been known to possess sedative, cardiovascular protective, anti-inflammatory, and antioxidant properties. Another secondary metabolite identified in H. capitellata is steroids. Plant sterols are essential for plant growth, reproduction, and response to various biotic and abiotic stresses. These potent phytosterols have long been used as cholesterol-lowering agents and have immunosuppressive and anti-inflammatory properties [11]. Steroids also act as electron donors, neutralizing free radicals and converting them into more stable compounds, thus providing the final chain reaction [12]. The present study also found tannins in the EEHC. These phytochemicals are crucial in the chemical defense mechanism of plants against pathogens and herbivores, as the bitter taste of tannins is less preferred by herbivores [11]. Tannins exhibit antioxidant properties, functioning by donating hydrogens and chelating metal ions such as Fe (II), Zn (II), Cu (II), and intervening in Fenton reactions [12]. Saponins are plant chemical compounds that exhibit soap-like characteristics and foam formation when mixed with water. When dissolved in water, saponins reduce the surface tension of water, allowing the formation of stable small bubbles. Due to their surfactant activity, saponins are excellent foaming agents (highly stable). In nature, plants rely on saponins as a mechanism to defend against parasites. Similarly, when consumed by animals, saponins provide similar protective abilities against harmful organisms [11]. Additionally, saponins are utilized by animals as natural antioxidants and also stimulate programmed cell death in tumor cell masses [12].
Polyphenols and flavonoids are important antioxidants and constitute a significant proportion in plants. The total phenolic and flavonoid content of H. capitellata is 67.85 mg GAE/g and 41.64 mg QE/g, respectively. According to a study published by Kähkönen et al. [21], plants with a polyphenol content greater than 20 mg GAE/g exhibit strong antioxidant activity. Phenols are essential in plants for growth, particularly in lignin and pigment synthesis. They also provide structural integrity and support for plant tissues. More importantly, plant phenolic phytoalexins are secreted by plants to repel or destroy microorganisms and certain pathogens. Phenolic compounds are widespread in plants, and when plant-based foods are consumed, these plant chemicals contribute to enhanced absorption of natural antioxidants [11]. Flavonoids are plant chemical compounds found in many plant species and possess several medicinal benefits, including anticancer, antioxidant, anti-inflammatory, antiviral, neuroprotective, cognitive-enhancing effects during aging, prevention of cardiovascular metabolic disorders, anticancer, and anti-proliferative properties. Extracts rich in flavonoids offer numerous medicinal benefits [22].
Evaluation of gastric acid secretion inhibition
Mice treated with indomethacin (IND group) exhibited elevated gastric acidity, as evidenced by a reduction in gastric pH, an increase in free acid content, and total acid levels compared to the distilled water group (DW) (Table 4, p<0.05). Omeprazole treatment significantly mitigated gastric acidity compared to the IND group (p<0.05), demonstrating the efficacy of omeprazole in acid control. All three doses of the extract (300, 350, and 400 mg/kg) markedly reduced gastric acidity compared to the IND-only group (p<0.05). Particularly noteworthy, in the group treated with the highest dose of the extract (400 mg/kg), the decrease in total acid content was nearly equivalent to that of the distilled water group (p>0.05) and significantly differed from the IND+OME group (p<0.05). Consequently, EEHC demonstrated a positive impact in reducing gastric acidity in mice, exhibiting efficacy even surpassing that of omeprazole. These results may provide support for further research and the development of novel approaches in the treatment of gastric acidity.
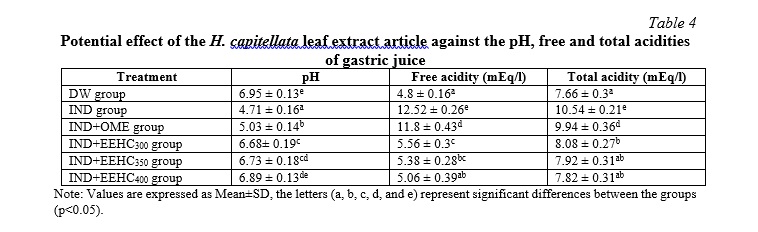
Most of the drugs currently used in the treatment of gastric ulcers, such as lansoprazole, pantoprazole, esomeprazole, omeprazole, etc., work by reducing gastric acid secretion. Acid suppression leads to an increase in blood gastrin levels. However, gastrin stimulates gastric acid secretion, increases enterochromaffin and carcinoid cell proliferation. Additionally, increased gastric acid levels increase the risk of intestinal infection and H. pylori infection. This creates favorable conditions for the development of gastric inflammation, gastric atrophy, and an increased risk of glandular stomach cancer and intestinal metaplasia. Moreover, increased gastric acid also reduces calcium absorption in the intestine, impairs vitamin B12 absorption, and increases the risk of osteoporosis and anemia [3]. The current study demonstrated that the gastroprotective effect of the ethanol leaf extract of H. capitellata did not significantly increase gastric acidity, which was significantly different from the effect of omeprazole (p<0.05). The non-significant increase in pH, free acid, and total acid in the stomach caused by the extract may be attributed to the moderate inhibitory effect of the extract components on gastric proton pump and histidine decarboxylase [3]. This provides an advantage for the extract over existing anti-ulcer medications.
Determination of gastric mucosal injury
The results presented in Fig. 2 reveal the impact of EEHC on the ulcer index and the percentage of ulcer inhibition. Indomethacin induced a significant elevation in the ulcer index (0.78±0.02) in mice. Notably, treatment with the extract demonstrated a substantial improvement in ulcer inhibition. EEHC at doses of 300, 350, and 400 mg/kg exhibited notable ulcer inhibition capacities (56.07%, 58.93%, and 63.93%, respectively), accompanied by ulcer indices (0.34±0.04, 0.32±0.03, and 0.28±0.02, respectively) that were significantly different from the IND group (0% ulcer inhibition, ulcer index 0.78±0.02) (p<0.05). The therapeutic effect of EEHC on gastric ulcers proved to be comparable to the effect of omeprazole (58.93% ulcer inhibition, ulcer index 0.32±0.02) (p>0.05). These findings suggest that the ulcer healing properties observed in the ethanol leaf extract of H. capitellata indicate its gastroprotective effects. The data underscore the potential of EEHC as a promising agent in mitigating indomethacin-induced gastric ulcers and emphasize its comparable efficacy to the standard medication, omeprazole.
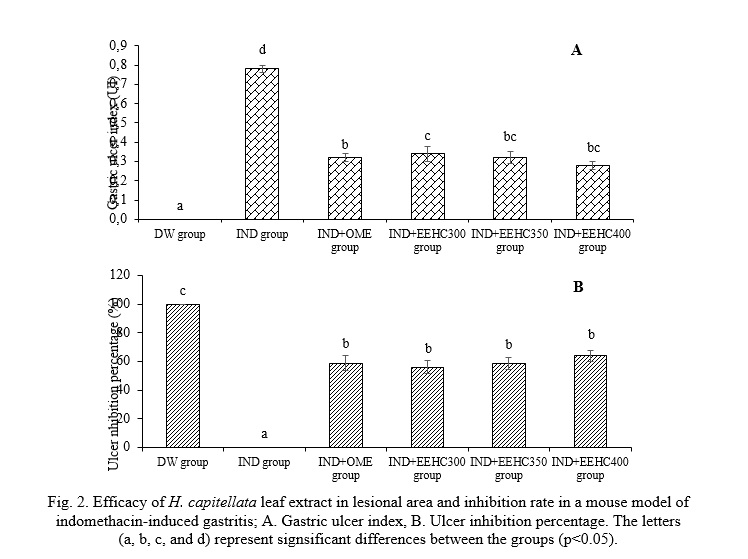
Gastric ulcer is a common digestive disorder characterized by inflammatory lesions or craters in the mucosal and submucosal layers, resulting from an imbalance between damaging factors such as acid, pepsin, H. pylori, and defensive factors such as gastric mucus, bicarbonate ions, and prostaglandins along with the innate resistance of the mucosal cells [4]. Currently, NSAIDs are widely used worldwide for their analgesic and anti-inflammatory properties. Indomethacin (IND), an NSAID, is widely used in clinical practice. However, IND can cause severe ulceration and erosion that extends to the gastric mucosa. IND induces gastric mucosal damage by activating inflammatory cells, producing pro-inflammatory cytokines, and inducing oxidative stress. The generation of free radicals and lipid peroxides plays a crucial role in the development of IND-induced gastric mucosal injury [16]. The ulcer index and percentage of ulcer inhibition data from this study indicate that EEHC has potential anti-inflammatory and antioxidant effects against IND-induced gastric mucosal damage. Plant components such as flavonoids, tannins, and saponins have been reported in several ulcer-protective studies and may contribute to gastric mucosal protection. Flavonoids and tannins are among the cell-protective agents that have been widely acknowledged for their ulcer-preventive efficacy. It is believed that these compounds can stimulate the secretion of mucus, bicarbonate, and prostaglandins, while counteracting the deleterious effects of oxidative substances in the gastrointestinal tract. Tannins can inhibit the development of ulcers through protein precipitation and their vasoconstrictive effects. Their ulcer-sealing effects may help in precipitating protein over the ulcer site, thus forming a water-impermeable layer on the mucosa, impeding the secretion from the intestine, and protecting the underlying mucosa from toxins and other irritants [2].
Hematology and biochemistry
Table 5 provides a comprehensive overview of the alterations in hematological and biochemical parameters observed in mice undergoing treatment for gastric inflammation induced by IND. A significant increase in WBC count was evident during the IND-induced gastric ulceration process, displaying a noteworthy difference compared to the DW group (p<0.05). Conversely, the RBC count demonstrated a significant decrease during IND treatment (p<0.05). The counts of various white blood cell types (LYM, MONO, and GRA) exhibited significant variations in the IND-treated groups compared to the DW group (p<0.05). The levels of ALP, CRP, and ESR significantly increased after mice were treated with IND (p<0.05). The reversal of these adverse effects was notable after treating the mice for gastric ulceration with EEHC and OME. Specifically, in the EEHC300, EEHC350, and EEHC400 groups, the WBC count, ALP, CRP, and ESR levels demonstrated a significant decrease compared to the IND group (p<0.05), while the RBC count significantly increased (p<0.05). These findings suggest that EEHC has a potential protective effect on hematological and biochemical parameters, counteracting the IND-induced alterations. The observed improvements in these parameters further support the potential therapeutic benefits of EEHC in the context of gastric ulceration.
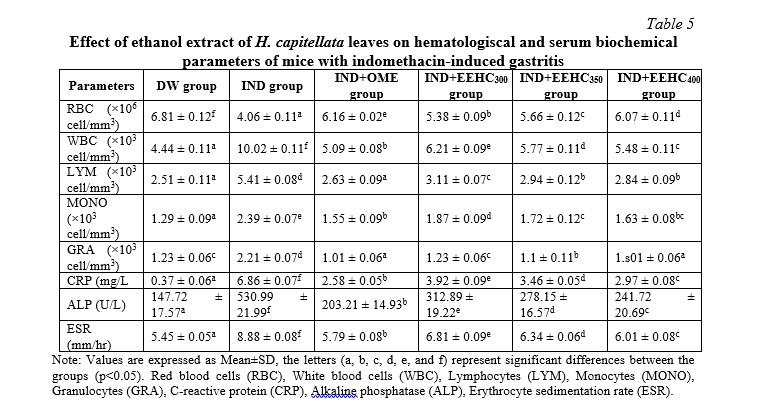
The treatment of gastric ulcers in animals depends on various factors, such as age, overall health status, disease severity, and drug absorption capacity. Physiological studies have shown that stress from any source can affect the endocrine, hematopoietic, and immune systems. The white blood cell count and neutrophil count increase, while lymphocyte, eosinophil, and monocyte counts decrease due to stress [23]. Red blood cells (RBCs) are crucial in the ulcer healing process as they are responsible for carrying oxygen to the affected tissue. The availability of oxygen affects all stages of the healing process, making it essential for successful healing. A decrease in RBC count due to aspirin use has also been previously observed in gastric tissue injury [24], which corresponds to the current study where RBC count decreased in mice treated with IND. In this study, the RBC count increased in mice treated with EEHC and OME, providing more oxygen for granulation tissue formation and faster wound healing. The body's cellular protective mechanisms are mobilized through biological reactions in the injured area, accompanied by vascular and nervous reactions, which are necessary for tissue repair. Previous studies have shown that the natural healing process in mice requires an enhanced immune response through increased production of white blood cells [23]. The current study observed an increase in WBC count in mice after inducing ulceration with IND, indicating an immune response in animals with IND-induced inflammation. Lymphocyte (LYM) and granulocyte (GRA) counts are indicative of the inflammatory status in animals. Recently, increased lymphocyte and granulocyte counts have been observed in inflammatory disorders such as splenitis and colitis. An increase in lymphocyte and granulocyte counts indicates a more pronounced inflammatory condition, while a decrease in these parameters indicates reduced inflammation, leading to improved healing ability [23]. In this study, as the healing process progressed, the mice maintained low lymphocyte and granulocyte counts, indicating a faster healing process. C-reactive protein (CRP) and alkaline phosphatase (ALP) play essential roles in tissue necrosis, and their levels increase in alcohol-induced, aspirin-induced, and IND-induced tissue injury [25]. During the healing process in the current study, the activities of these parameters decreased in mice treated with the extract. The observed reduction may be a mechanism associated with enhanced ulcer healing reported in previous studies [26].
Changes in malondialdehyde (MDA) levels in gastric mucosa
The presented results in Fig. 3 highlight the concentration of malondialdehyde (MDA) in the gastric mucosa and its response to different treatments. The concentration of MDA in the distilled water (DW) group was measured at 2.39±0.19 nM/g tissue, establishing a baseline for comparison. A substantial and statistically significant increase in MDA concentration was observed in the gastric mucosa of the IND group (12.54±0.23 nM/g tissue) compared to the DW group (p<0.05). This elevation indicates oxidative stress or lipid peroxidation in response to IND-induced gastric inflammation. Mice treated with EEHC at doses of 300, 350, and 400 mg/kg showed varying levels of MDA (7.72±0.13, 6.82±0.14, and 5.29±0.15 nM/g tissue, respectively). Although EEHC at 300 and 350 mg/kg did not significantly reduce MDA levels compared to the IND group (p>0.05), treatment with EEHC at 400 mg/kg resulted in a significant decrease in MDA concentration, approaching the level observed in the IND+OME group (p<0.05), suggesting a dose-dependent antioxidant effect. This suggests a potential dose-dependent response, with higher doses of EEHC showing a trend toward mitigating the elevated MDA levels induced by IND. The ratios presented do not indicate a significant overall improvement in MDA concentration in the gastric tissue of the EEHC-treated groups, as mentioned in Fig. 3. So, while the lower doses of EEHC did not show significant improvement in MDA concentration, the higher dose (400 mg/kg) demonstrated a trend toward normalization, suggesting a potential dose-dependent effect.
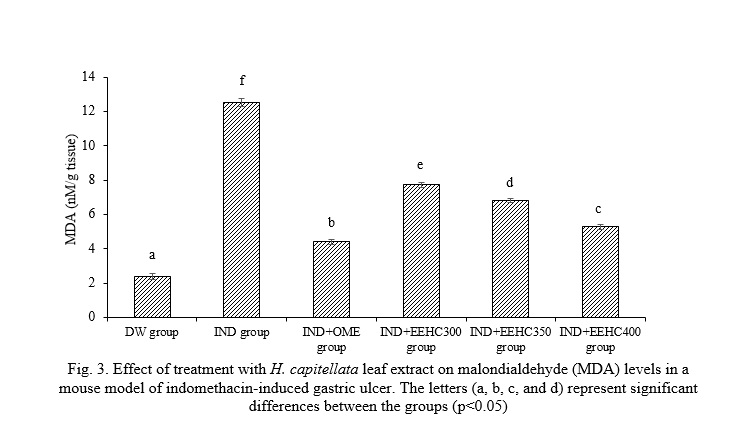
Oxidative damage becomes more pronounced with the process of lipid peroxidation, which is crucial in the pathophysiology of gastric injury involving cellular and membrane damage. In previous studies, the amount of lipid peroxidation has been shown to be associated with IND-induced gastric mucosal damage [16]. The increase in MDA levels is correlated with tissue damage and is an important factor in NSAID-related gastric injury [27]. The permeation of IND into the subepithelial layer causes tension in the gastric tissue, thus leading to gastric ulceration. The production of ROS and oxidative stress are the main causes of increased lipid peroxidation (MDA). The current study confirmed the findings of Abbas and Sakr and Fahmy et al. in observing an increase in the levels of the end products of lipid peroxidation, malondialdehyde (MDA), in gastric tissues after IND treatment [28, 29]. The level of MDA is believed to reflect cellular membrane damage through the intermediate of free radicals [29]. As demonstrated in the results of the present study, the treatment of gastric ulcers by EEHC and omeprazole significantly reversed the changes induced by IND in MDA levels, suggesting that the mechanism of both the anti-inflammatory effects of omeprazole and EEHC may involve their antioxidant effects.
The activity of antioxidants in the stomach
The results presented in Table 6 offer insights into the impact of EEHC on antioxidant enzymes in the context of gastric mucosal response to IND. The activities of CAT and MPO were significantly inhibited in the gastric mucosa of animals exposed to IND, indicating a reduction in antioxidant defenses (p<0.05). The values for CAT (34.85±0.11 mM/min/mg of tissue) and MPO (9.18±0.11 mM/min/mg of tissue) in the IND group were markedly higher compared to the intact gastric mucosa (CAT: 17.55±0.15, MPO: 2.52±0.12 mM/min/mg of tissue, respectively). Pretreatment with EEHC mitigated the decline in CAT and MPO activities induced by IND, with the 400 mg/kg EEHC group showing the most significant improvement (CAT: 24.95±0.14 mM/min/mg of tissue, MPO: 4.56±0.12 mM/min/mg of tissue) compared to the IND-only group (p<0.05). IND led to a decrease in GSH (2.57±0.09 nM/mg tissue) and TAC (11.93±0.12 nM/mg protein) compared to the control group (p<0.05). The control group had higher GSH (5.32±0.11 nM/mg tissue) and TAC (29.18±0.15 nM/mg protein) values. Pretreatment with EEHC increased GSH and TAC activities in the stomach, with the most notable efficacy observed at a dose of 400 mg/kg (GSH: 4.51±0.06 nM/mg protein, TAC: 22.82±0.07 nM/mg protein) (p<0.05). Therefore, EEHC demonstrated a protective effect on antioxidant enzymes, including CAT, MPO, GSH, and TAC, in the gastric mucosa exposed to IND-induced inflammation. The most pronounced improvements were observed at the highest dose of EEHC (400 mg/kg), indicating a dose-dependent response. These findings suggest that EEHC may play a crucial role in preserving the antioxidant balance in the gastric mucosa under inflammatory conditions induced by IND.
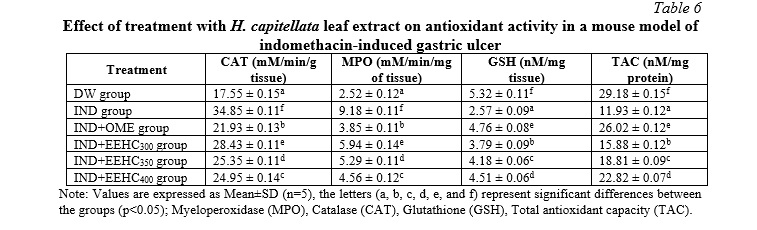
Reactive oxygen species (ROS) are generated during cellular metabolism under both physiological and pathological conditions. However, excessive ROS release is evident in disease states [20]. Organisms protect against the adverse effects of ROS through specific antioxidant defense systems, including enzymatic and endogenous scavenging systems. Specifically, catalase (CAT) and myeloperoxidase (MPO) are characterized as the first line of defense against oxidative stress. MPO can catalyze the conversion of hydrogen peroxide (H2O2) to hypochlorous acid (HOCl) in the presence of chloride ions, generating reactive oxygen species (ROS) that directly damage pathogens. CAT neutralizes the harmful effects of H2O2 by converting it into water. When excessive ROS is released, these protective mechanisms can become overwhelmed. IND has been shown to decrease the activity of CAT and MPO [30], which is also confirmed in the present study. Furthermore, we demonstrated that treatment with EEHC modulated the increased levels of CAT and MPO in the IND-induced gastric ulcer animal model. The elevation of CAT and MPO activities will prevent lipid peroxidation and tissue destruction, thus promoting the recovery of IND-induced gastric damage [20]. Glutathione (GSH) and total antioxidant capacity (TAC) protect cells against free radicals, peroxides, and other harmful compounds. Reduced GSH and TAC levels in the gastric mucosa can lead to the accumulation of free radicals, which may cause cellular membrane damage through lipid peroxidation [29]. This is consistent with our study, which reported a decrease in GSH and TAC concentrations in the gastric tissue of mice treated with IND. Gastric injury depletes GSH and TAC, which serve as the first line of defense for cells against oxidative damage. This can lead to more severe tissue damage during gastric ulceration. Treatment with OME and EEHC restored the levels of GSH and TAC, indicating a potential protective effect against oxidative damage.
Effect of EEHC on the release of inflammatory cytokiness
The findings presented in Table 7 highlight the impact of H. capitellata leaf extract on inflammatory markers in the context of IND-induced inflammation. Administration of IND significantly increased the expression of inflammatory markers, including IL-1β, IL-6, and TNF-α (p<0.05). Additionally, there was a significant reduction in the concentration of anti-inflammatory cytokine IL-10 compared to the DW group (p<0.05). Treatment with H. capitellata leaf extract at doses of 300, 350, and 400 mg/kg resulted in a reversal of these IND-induced effects (p<0.05). The concentrations of IL-1β, IL-6, TNF-α, and IL-10 were significantly decreased, indicating a reduction in the inflammatory response (p<0.05). Notably, the concentration of IL-10 was significantly increased, suggesting an enhancement of anti-inflammatory mechanisms (p<0.05). These results suggest that H. capitellata leaf extract possesses anti-inflammatory properties, as evidenced by its ability to modulate the expression of pro-inflammatory cytokines (IL-1β, IL-6, TNF-α) and enhance the concentration of the anti-inflammatory cytokine IL-10. The observed effects at various doses of the extract indicate a dose-dependent response, with higher doses exhibiting more pronounced anti-inflammatory effects. Overall, these findings support the potential therapeutic use of H. capitellata leaf extract in mitigating inflammatory responses, providing a basis for further exploration in the development of anti-inflammatory interventions.
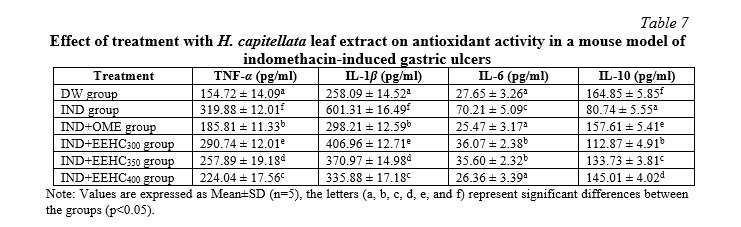
In the pathogenesis of gastric ulcer, mucosal damage plays a crucial role mediated by certain pro-inflammatory cytokines synthesized in the gastric mucosa upon IND administration. Some important cytokines involved include TNF-α, IL-6, IL-1β, and IL-10, released from macrophages during inflammation [31]. Indomethacin induces high-grade mucosal inflammation, characterized by a significant increase in infiltration of inflammatory cells in the gastric mucosa, elevated levels of TNF-α, IL-1β, IL-6, and reduced levels of IL-10 compared to the DW group. Treatment with the plant extract at all three doses inhibited infiltration of gastric inflammatory cells, significantly decreased the levels of TNF-α, IL-6, IL-1β, and increased the level of IL-10 in the serum. This indicates the anti-inflammatory role of EEHC. IL-6 stimulates lymphocytes, macrophages, and neutrophils at the site of inflammation, leading to excessive production of ROS and lysosomal enzymes, resulting in tissue damage in gastric ulcers. TNF-α plays a crucial role in IND-induced mucosal damage by reducing blood flow to the gastric mucosa and upregulating the expression of gastrin and vascular endothelial growth factor in the gastric mucosa, thereby hindering the ulcer healing process. TNF-α also activates the NF-κB pathway as an intermediate for the transcription process of certain adhesion molecules, leading to the infiltration of inflammatory cells observed in mice treated with IND [3]. IL-1β is a pro-inflammatory cytokine produced during gastric inflammation and enhances the inflammatory response, causing an imbalance between detrimental and defensive factors such as increased angiogenesis, increased mucous membrane permeability, and enhanced infiltration of inflammatory cells. IL-10 is an anti-inflammatory cytokine that can reduce the inflammatory response, maintain a balance between inflammation and anti-inflammation by inhibiting the production of pro-inflammatory cytokines. IL-10 helps reduce mucosal damage, alleviate inflammation, dampen excessive immune responses, and inhibit the proliferation of inflammatory cells [16]. In the present study, the decrease in TNF-α, IL-1, IL-6 levels, and the significant increase in IL-10 levels in serum indicate a reduction in the oxidative and inflammatory conditions. A previous study suggested that the anti-inflammatory effects of the extract are mediated through the downregulation of cytokines such as TNF-α, IL-1, and IL-6 [32]. The authors explained that this improvement is attributed to the polyphenolic content of the extract, which possesses strong antioxidant, anti-inflammatory, and tissue-repairing properties.
Macroscopic and histopathologic evaluation
The visual observations presented in Fig. 4 provide valuable insights into the protective effects of EEHC and OME against gastric mucosal damage induced by IND. The distilled water control group (Fig. 4Aa) did not exhibit mucosal ulcerations or bleeding lesions, indicating a healthy gastric mucosa. In contrast, IND-treated mice displayed distinct localized ulcerative and hemorrhagic lesions in the gastric mucosa (Fig. 4Ab). This observation aligns with the expected effects of IND, causing visible damage to the gastric lining. Mice treated with EEHC and OME showed a marked inhibition of total gastric lesions (Fig. 4Ac, 4Ad). The mucosal lesions decreased in a dose-dependent manner, suggesting a potential protective effect of both EEHC and OME against IND-induced gastric mucosal damage. The most pronounced inhibitory effect on the increased area of gastric mucosal lesions caused by IND was observed in the EEHC-treated group at a dose of 400 mg/kg (Fig. 4Ac). This indicates a potential dose-dependent response, with higher doses of EEHC providing greater protection against mucosal damage. Overall, these visual observations corroborate the biochemical and molecular findings, suggesting that EEHC has a protective effect against IND-induced gastric ulcerations and lesions. The dose-dependent response and the comparative efficacy with OME further support the potential therapeutic value of EEHC in preventing or mitigating gastric mucosal damage induced by inflammatory agents such as IND.
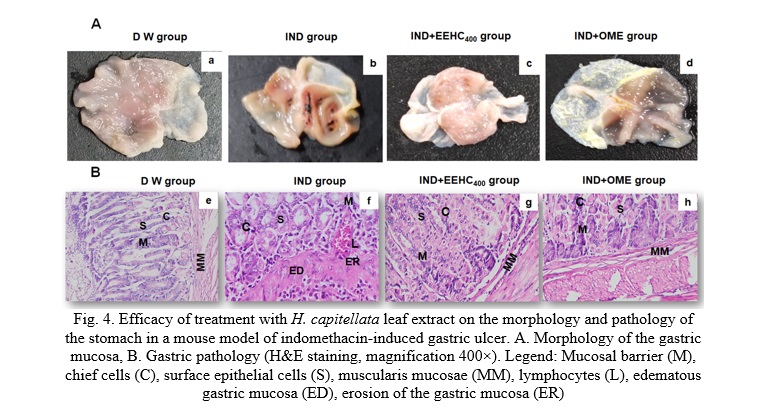
The detailed histological observations in Fig. 4B provide crucial insights into the protective effects of H. capitellata leaf extract (EEHC) against severe gastric mucosal injuries induced by IND. Severe mucosal edema and erosion, extensive localized surface mucosal injury, infiltration of lymphocytes, deep infiltration of inflammatory cells, erosion of the muscular layer resulting in congestion or hemorrhage, and necrotic ulcerative lesions in the gastric glands were observed after IND treatment (Fig. 4Bf). These findings illustrate the damaging impact of IND on the gastric mucosa. Treatment with H. capitellata leaf extract (Fig. 4Bg) significantly suppressed the IND-induced gastric mucosal injuries. Notable observations include the process of surface epithelial cell regeneration, a significant reduction in the infiltration of inflammatory cells, restoration of muscular layers, and the absence of mucosal erosion. The mucosal recovery expression in the IND+EEHC400 group (Fig. 4Bh) was comparable to the IND+OME group. This suggests that EEHC, particularly at a dose of 400 mg/kg, exhibited a level of mucosal recovery comparable to the standard medication omeprazole (OME). The mucosal recovery in the IND+EEHC400 group showed no significant difference compared to the distilled water control group (Fig. 4Be). This indicates that the protective effects of EEHC were successful in mitigating the severe mucosal injuries induced by IND, restoring the gastric mucosa to a condition similar to the untreated control group. So, these histological observations provide compelling evidence of the gastroprotective effects of H. capitellata leaf extract against severe gastric mucosal injuries induced by IND. The regenerative processes and the absence of significant differences compared to the control group suggest that EEHC has the potential to aid in the recovery and repair of gastric mucosa subjected to inflammatory insults.
Histological observations demonstrated the regeneration of gastric mucosal epithelium in mice treated with EEHC and OME. According to Ajayi et al., the regeneration of ulcer bed mucosal epithelium is a crucial process for healing gastrointestinal mucosal injuries [23]. Without the restoration of a continuous mucosal barrier, the mucosa becomes susceptible to mechanical, chemical, and infectious damage, thereby impeding the ulcer healing process. Previous reports have also shown that reducing the infiltration of lymphocytes into ulcerated gastric tissues is associated with promoting the healing of chronic ulcers caused by IND in mice [33]. The findings of the present study are consistent with these previous findings.
Conclusions. This study validates the efficacy of H. capitellata leaf ethanol extract (EEHC) in alleviating gastric mucosal damage. EEHC demonstrated a comprehensive impact, including reduced ulcer index, decreased white blood cell (WBC) count, lowered malondialdehyde (MDA) concentration, and modulation of oxidative enzymes (myeloperoxidase and catalase). Significant changes were also observed in pro-inflammatory cytokines (TNF-α, IL-6, IL-1β), increased red blood cell (RBC) counts, and enhanced endogenous antioxidants (glutathione and total antioxidant capacity). Notably, the anti-inflammatory cytokine IL-10 increased compared to the indomethacin-treated group. Histological analysis revealed gastric mucosal epithelial regeneration with EEHC treatment. The study explored active compounds, predicted targets, and mechanisms of EEHC's anti-inflammatory actions through pharmacological analysis. These effects primarily involved reinforcing the antioxidant defense system and modulating inflammatory markers. In conclusion, H. capitellata leaf extract exhibits distinctive anti-inflammatory properties, showing promise for novel plant-based therapeutic strategies in managing gastric ulcer inflammation and serving as a potential source of medicinal ingredients for further research.
Financial support
No financial support has been provided for this work.





















Reference lists